Exosomes Stand Out as Potential Blood Biomarkers
Quick Links
Exosomes—small membrane-bound packets of cellular components that escape the cell—are coming under suspicion as players in neurodegenerative disease. Could they yield a biomarker? At the Alzheimer’s Association International Conference held July 12-17 in Copenhagen, Denmark, two researchers argued that exosomes found in blood hold the potential to signal disease. One looked at Alzheimer’s disease proteins packaged inside exosomes, the other examined the microRNAs therein. Both researchers reported ways to tell patients from healthy controls, and perhaps even predict future disease.
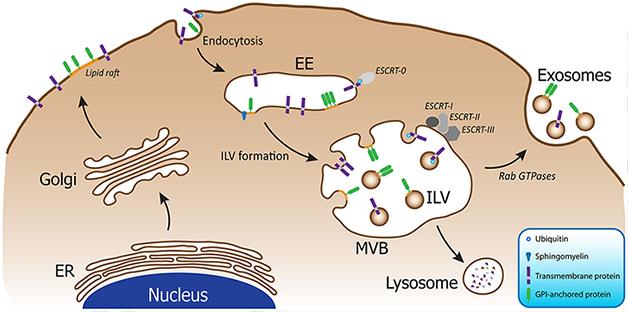
Multivesicular bodies (MVBs) fuse with the plasma membrane, releasing their inner vesicles as exosomes. [Image courtesy of Bellingham et al., 2012.]
Within the cell’s complex endosomal trafficking system, late endosomes called multivesicular bodies (MVBs) can either fuse with lysosomes for breakdown of their components, or merge with the plasma membrane and spill their intraluminal vesicles outside the cell (see image above). In the latter case, these intraluminal vesicles become known as exosomes. Containing tiny amounts of the cell’s cytoplasm, these packets essentially represent a sample of the intracellular environment, and are found in the cerebrospinal fluid (CSF), blood, saliva, urine, and milk. Previous studies have reported that exosomes contain proteins associated with neurodegenerative diseases, including Aβ, α-synuclein, and tau (see Rajendran et al., 2006; Emmanouilidou et al., 2010; and Saman et al., 2011). Given that exosomes have found some success as diagnostic biomarkers in cancer, some researchers wonder if they might also lead to a noninvasive biomarker of neurodegenerative disease (for a review, see Properzi et al., 2013).
Dimitrios Kapogiannis, National Institute on Aging, Bethesda, Maryland, generated excitement among attendees of an AAIC session when he presented new findings suggesting such a possibility for AD. He and colleagues isolated exosomes from the blood of 57 patients diagnosed with probable AD and 57 age- and sex-matched controls. The scientists enriched for exosomes from neural cells with antibodies for the neuronal adhesion proteins L1 and NCAM. While not expressed exclusively by neurons, these transmembrane proteins helped concentrate neuronal exosomes in the sample. The researchers then centrifuged the samples and used ELISA to quantify their levels of Aβ42, total tau (t-tau), and tau phosphorylated at T181 (ptau-181) or S396 (ptau-396).
Kapogiannis reported that the AD patients had significantly higher levels of Aβ42, ptau-181, and ptau-396 in their blood-based exosomes. Individually, the levels of each protein predicted AD with a percent accuracy in the high 90s. Combined, the three proteins’ predictive ability approached 100 percent, meaning they distinguished AD patients from controls with almost no overlap. T-tau yielded no difference between groups.
To see if the same approach could predict whether cognitively normal people would progress to dementia, Kapogiannis and colleagues sampled 24 patients who gave a blood sample at AD diagnosis and had already given another 1-10 years prior. These were matched with 24 more controls. All three proteins were elevated in early blood samples from those who later developed AD. While phosphorylated tau had already reached levels seen in diagnosed AD, Aβ42 measurements at these prediagnosis time points were intermediate between controls and patients, suggesting the peptide correlates with disease progression.
Together, the three proteins “near-perfectly discriminated between Alzheimer’s patients and controls,” Kapogiannis said. “There is also a possibility that these exosomal markers may … be used to predict disease at the preclinical stage.” Different proteins in exosomes could identify other neurodegenerative diseases, he pointed out.
Audience members lined up to ask questions after the presentation. One person wanted to know whether exosomes not enriched for neuronal origin gave a similar result. Kapogiannis said group differences were measurable even without selecting for neuronal markers, but were less distinct. Someone else asked whether the researchers planned to confirm these findings in exosomes from CSF, which would reflect neuronal sources even better. Kapogiannis responded that this had not been done yet because a high volume of CSF was required to obtain enough exosomes. A third listener asked about truncated tau in the exosomes, but Kapogiannis has not looked beyond those four proteins yet.
“That this test works in blood is encouraging,” said Reisa Sperling, Brigham and Women’s Hospital, Boston. She found the prognostic potential most intriguing, as it could make the test useful for the Anti-Amyloid Treatment in Asymptomatic Alzheimer’s (A4) study to predict who has pathology. “To have a blood test that correlates with lumbar puncture results or PET imaging data would be an incredible breakthrough,” Sperling said, adding that it will be important to replicate the study in a larger sample of people characterized by biomarkers.
“The results were startling,” said Ralph Nixon, NYU Langone Medical Center, New York. “The degree of separation between AD and control and the ability to detect changes years before conversion was impressive.” He cautioned that the process of extracting exosomes and enriching for ones from neural cells is time-consuming for a technique that might be applied on a wide scale, but called the technical challenges surmountable. In the future, researchers might be able to refine the enriching marker to one that distinguishes neurons specifically, or even particular neuronal populations, he suggested. In addition, exosomal protein levels might hold clues to how disease disrupts the endosomal pathway, he said.
Researchers are also examining other exosome components as potential biomarker sources for Alzheimer’s. Andrew Hill, University of Melbourne, Australia, is examining the utility of microRNAs (miRNAs). These could be easier to detect than proteins because they can be amplified by PCR and do not require special antibodies, he said.
Hill and colleagues isolated exosomes from blood serum samples of 23 AD patients, three people with mild cognitive impairment (MCI), and 23 healthy controls from the Australian Imaging Biomarkers and Lifestyle (AIBL) study. Extracting and sequencing the RNA, the researchers identified 1,419 miRNAs, 220 of which showed up in all the samples. Of those, 14 were upregulated and three downregulated in AD and MCI compared with controls. A machine-learning technique called random forest modeling identified the 16 most predictive ones. They related to APP processing, apoptosis, and endoplasmic reticulum stress, Hill said.
To see if this set could predict disease, Hill’s group tested 15 more patients clinically diagnosed with AD as well as 35 healthy controls. All had undergone positron emission tomography with Pittsburgh Compound B (PiB-PET) to detect amyloid accumulation. Combined with information about age, sex, and ApoE4 status, the miRNA profile correctly diagnosed 13 of the 15 patients. It also correctly picked out 27 of 35 healthy controls, though five of the incorrectly characterized controls in fact had high amyloid burden. The researchers will wait and see if these five progress to AD in longitudinal assessments. If they do, it would mean the test has a specificity of 91 percent, he said.
Hill said he plans to examine a larger number of patients and validate the test in different cohorts, including more people with MCI. He will also check to see if the miRNA signature correlates with PiB-PET imaging. If so, the miRNA signature could become a low-cost, noninvasive screen for amyloid pathology. To extend the technique to other diseases, his group is looking at exosomes in people with Parkinson’s as well as animal models of other neurodegenerative disorders.
“This method has potential,” said Robert Nagele, Rowan University, Stratford, New Jersey, who chaired the session. The deep RNA sequencing makes it labor-intensive and expensive for now, but these are early days. Once the researchers settle on reliable biomarkers, they can streamline the method and develop simpler, cheaper assays, Nagele said. He noted that this method requires combination with clinical parameters to be highly accurate in a clinical situation. However, he said that for presymptomatic tests, it would be even better to find biomarkers that can stand on their own.
Exosomes appear to contain yet more proteins related to neurodegenerative disease. Markus Otto, University of Ulm, Germany, presented on a recent paper (see Feneberg et al., 2014), in which he and colleagues reported fragments of TDP-43 in exosomes from CSF. Quantifying these fragments in exosomes, or possibly phosphorylated and aggregated forms of the protein, may help diagnose TDP-43-related disorders such as amyotrophic lateral sclerosis and frontotemporal lobar degeneration in the future, he wrote to Alzforum in an email.—Gwyneth Dickey Zakaib
References
Paper Citations
- Rajendran L, Honsho M, Zahn TR, Keller P, Geiger KD, Verkade P, Simons K. Alzheimer's disease beta-amyloid peptides are released in association with exosomes. Proc Natl Acad Sci U S A. 2006 Jul 25;103(30):11172-7. PubMed.
- Emmanouilidou E, Melachroinou K, Roumeliotis T, Garbis SD, Ntzouni M, Margaritis LH, Stefanis L, Vekrellis K. Cell-produced alpha-synuclein is secreted in a calcium-dependent manner by exosomes and impacts neuronal survival. J Neurosci. 2010 May 19;30(20):6838-51. PubMed.
- Saman S, Kim W, Raya M, Visnick Y, Miro S, Jackson B, McKee AC, Alvarez VE, Lee NC, Hall GF. Exosome-associated tau is secreted in tauopathy models and is selectively phosphorylated in cerebrospinal fluid (CSF) in early Alzheimer's Disease. J Biol Chem. 2011 Nov 4; PubMed.
- Properzi F, Logozzi M, Fais S. Exosomes: the future of biomarkers in medicine. Biomark Med. 2013 Oct;7(5):769-78. PubMed.
- Feneberg E, Steinacker P, Lehnert S, Schneider A, Walther P, Thal DR, Linsenmeier M, Ludolph AC, Otto M. Limited role of free TDP-43 as a diagnostic tool in neurodegenerative diseases. Amyotroph Lateral Scler Frontotemporal Degener. 2014 Sep;15(5-6):351-6. Epub 2014 May 16 PubMed.
External Citations
Further Reading
Papers
- Lee Y, El Andaloussi S, Wood MJ. Exosomes and microvesicles: extracellular vesicles for genetic information transfer and gene therapy. Hum Mol Genet. 2012 Oct 15;21(R1):R125-34. Epub 2012 Aug 7 PubMed.
- Dinkins MB, Dasgupta S, Wang G, Zhu G, Bieberich E. Exosome reduction in vivo is associated with lower amyloid plaque load in the 5XFAD mouse model of Alzheimer's disease. Neurobiol Aging. 2014 Aug;35(8):1792-800. Epub 2014 Feb 15 PubMed.
- Cheng L, Quek CY, Sun X, Bellingham SA, Hill AF. The detection of microRNA associated with Alzheimer's disease in biological fluids using next-generation sequencing technologies. Front Genet. 2013;4:150. PubMed.
- Bellingham SA, Guo BB, Coleman BM, Hill AF. Exosomes: vehicles for the transfer of toxic proteins associated with neurodegenerative diseases?. Front Physiol. 2012;3:124. Epub 2012 May 3 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
NanoSomix, Inc.
Dr. Nixon observed that a process that involves exosomes can be a time-consuming technique. While I generally agree with Dr. Nixon, I have been working with exosome isolation and characterization for some time and have recently developed a unique high-throughput process that makes such assays routine and consistent. NanoSomix, who owns the intellectual property of the assay described in Dimitrios Kapogiannis' presentation at AAIC, is in the final stages of validating this method and will be offering a laboratory developed test (LTD) for exosome surface marker CD81, Aβ1-42, total tau, and two forms of phospho-tau in the fall. I am happy to discuss our progress with anyone who is interested. Masato Mitsuhashi, M.D., Ph.D., NanoSomix, Inc. mmitsuhashi@nanosomix.com
Make a Comment
To make a comment you must login or register.