Paris: Renamed ARIA, Vasogenic Edema Common to Anti-Amyloid Therapy
Quick Links
After disappointing trial results for Alzheimer’s disease (AD) drugs, all eyes are on the Phase 3 clinical trials of two monoclonal antibodies to amyloid-β: bapineuzumab, developed by Janssen Alzheimer Immunotherapy and Pfizer, and solanezumab, by Eli Lilly and Company. We will have to hold our collective breath a little longer for the results, which should be available at around the same time for both trials.
Although there were several bapineuzumab presentations at the 2011 Alzheimer’s Association International Conference (AAIC, formerly ICAD), held 16-21 July in Paris, France, none shed new light on the all-important efficacy front. They did, however, suggest (for those looking for some good news) that the ongoing Phase 3 trials are unlikely to be derailed by safety concerns. In the process, they recast what was earlier thought to be a potentially show-stopping side effect of amyloid-targeting therapies into one that actually indicates the therapy is working.
When given intravenously to patients with mild to moderate Alzheimer’s disease, bapineuzumab has been shown by PET to decrease brain amyloid (see ARF related news story on Rinne et al., 2010). But the treatment also raised concern when some patients enrolled in the Phase 2 trial developed vasogenic edema—an abnormal accumulation of fluid—and tiny hemorrhages in the brain (see ARF related news story on Salloway et al., 2009 and ARF related news story).
At AAIC, scientists reported results of a re-evaluation of the Phase 2 safety data of bapineuzumab and of an ongoing long-term extension trial of a subset of patients enrolled in the Phase 2 trial. They revealed that these abnormalities are widespread among patients receiving bapineuzumab, but they appear “manageable,” according to Stephen Salloway of Butler Hospital and Brown University in Providence, Rhode Island, one of the clinical investigators involved in the bapineuzumab trials. “What we have learned about dosing and risk factors is helping us manage the vascular edema,” Salloway told ARF. “We have found that most cases are relatively mild and transient. Most people with vascular edema can be re-dosed and can then resume treatment. So this is something we can manage.”
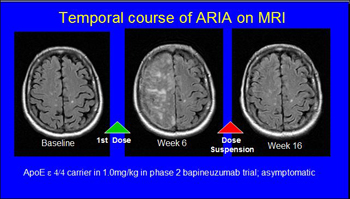
Series of MR scans of an AD patient who developed temporary ARIA without clinical symptoms while on bapineuzumab. View larger image. Image credit: Stephen Salloway and Reisa Sperling
If Not Music, Then What Is "ARIA"?
A working group of academic and industry experts, established by the Alzheimer’s Association to help guide the conduct of clinical trials of amyloid-lowering treatments for AD, renamed abnormalities corresponding to vasogenic edema and microhemorrhages, which are typically detected on MRI scans, as amyloid-related imaging abnormalities (ARIA)-E and ARIA-H, respectively (Sperling et al., 2011). “Vasogenic edema has radiological connotations of being tumor and abscess related,” said Salloway in a presentation on Thursday, 21 July, at AAIC. Changing the name would help identify the condition as a distinct phenomenon associated with AD and cerebral amyloid angiopathy (CAA)—the deposition of amyloid-β on the walls of blood vessels in the brain.
Microhemorrhages (or ARIA-H) occur as a result of healthy aging and in AD patients. In a presentation at AAIC, Meike Vernooij of Erasmus Medical Center in Rotterdam, The Netherlands, estimated that such abnormalities may be found in 3 to 15 percent of healthy older adults and in 18 to 32 percent of patients with AD. “The wide range of estimates reflects the fact that it depends on what tools and techniques you use,” said Vernooij. Although prevalence figures are unavailable for ARIA-E, some studies presented at AAIC suggest that it might also occur in some AD patients in the absence of treatment.
Although ARIA may occur “naturally” in some AD patients, there is no doubt that medications such as bapineuzumab hasten its development—and scientists now have some mechanistic hints as to why. At AAIC, Sally Schroeter and Wagner Zago, two researchers at Janssen Alzheimer Immunotherapy, presented a poster suggesting that bapineuzumab treatment causes blood vessels to become leaky due to the movement of amyloid-β from plaques in the brain into and then out of blood vessel walls. When 15–21-month-old PDAPP transgenic mice (see PDAPP line109) were treated weekly with a murine form of bapineuzumab for up to 36 weeks, blood vessel walls became temporarily weakened precisely at sites where Aβ had been deposited. During early stages of treatment, amyloid-β moves from the parenchyma to blood vessel walls. As Aβ is then removed from blood vessels, fluid leaks out. Aβ moves to the capillaries, where “it reduces the function of the water channel aquaporin AQ4,” Zago told ARF. “But eventually, the normal vascular integrity is restored.”
These results may explain how the side effects seen in AD patients treated with bapineuzumab occur. In his presentation, Salloway showed MRI results from one AD patient who participated in the Phase 2 bapineuzumab trial. In those images, the location of ARIA-E corresponded precisely to sites with high amounts of amyloid deposition. A follow-up MRI in the same patient showed that both the ARIA-E and amyloid were gone.
ARIA by the Numbers
The Phase 2 trial of bapineuzumab was an 18-month-long, double-blind, placebo-controlled, multiple ascending dose trial of 234 patients randomized to receive one of four doses of drug by intravenous infusion every 13 weeks. (It was carried out by the pharmaceutical companies Elan and Wyeth, but since then, Janssen Alzheimer Immunotherapy, a newly formed subsidiary of Johnson & Johnson, purchased Elan’s AD immunotherapy program and Pfizer bought Wyeth.)
Reisa Sperling of Brigham and Women's Hospital, Harvard Medical School, one of the clinical investigators involved in the Phase 2 bapineuzumab trial, presented results at AAIC 2011 of a re-evaluation of more than 2,000 MRI scans taken during the trial. “We realized the ability to recognize ARIA-E has evolved over time, so we suspected there might have been some missed cases,” said Sperling. “We also wanted to do a more systematic analysis of the data.”
The researchers identified 36 people (about 17 percent of the 210 trial participants who received treatment) whose MRI scans showed ARIA-E that the scientists thought was brought on by bapineuzumab; in other words, the abnormalities were absent at baseline and detected post-treatment. In 21 of those patients, ARIA-E had been identified during the Phase 2 trial. The additional 15 people with ARIA-E identified by Sperling’s re-evaluation had escaped detection in the original trial. “They had more subtle MRI changes, which may explain why they were missed,” said Sperling. None of these 15 patients had symptoms related to the ARIA-E, which can include headaches, loss of coordination, and disorientation. Many of the patients with ARIA-E (17 out of 36) also had ARIA-H that occurred during the trial (incident ARIA-H), indicating that the two conditions are related; incident ARIA-H was detected in only seven of the patients who did not have ARIA-E.
Although the prevalence of ARIA-E is high among patients receiving bapineuzumab, the fact that many patients did not have clinical symptoms related to the condition is reassuring to Sperling. “Those people continued to be treated throughout the trial because the ARIA-E was not detected, and they remained asymptomatic,” said Sperling. “I think that stopping therapy for patients who show evidence of ARIA-E makes sense. But on the other hand, if there are very mild cases and they get missed, we know from these data that we are probably not causing terrible harm to patients.”
Consistent with earlier studies, Sperling’s re-evaluation of the MRI data showed that patients with the apolipoprotein E4 allele (ApoE4) and those receiving the highest dose (2 mg/kg) of bapineuzumab were more likely to show evidence of ARIA-E. “There are no surprises here,” said Sid Gilman at the University of Michigan, commenting on an abstract of Sperling’s presentation. (The 2 mg/kg dose was dropped in the Phase 3 trials to minimize ARIA-E. For more information, see ARF related news story).
There were also few surprises in the interim results, presented by Salloway, on the ongoing long-term open-label extension study of 194 patients who continued to receive bapineuzumab after they completed the blinded Phase 2 trial. (Some subjects in the study who were originally receiving 2.0 mg/kg of the drug were switched to 1.0 mg/kg in the extension trial.) At the time of Salloway’s presentation, 94 people had received bapineuzumab treatment for at least three years, 43 for at least four years, and 22 for at least five years. The extension trial is expected to continue until the Phase 3 results are in.
Most participants (95 percent) reported a wide array of adverse events, including falls, agitation, urinary tract infections, upper respiratory tract infections, and anxiety. About 24 percent of patients reported adverse events that Salloway and colleagues tied to bapineuzumab treatment. The most common of those was ARIA-E, which occurred in 9.3 percent of patients. “The most encouraging result from this long-term study is that there are no new safety issues,” said Salloway.
Among the 18 patients with ARIA-E, two had headaches and other symptoms related to this condition, while the rest were asymptomatic. In addition, the risk of developing ARIA-E diminished as people received more infusions of the drug. The cumulative risk of developing ARIA-E dropped from 6.7 percent for infusions 1 to 3 to 2.7 percent for infusions 4 to 10. “This result,” said Salloway, “sheds light on the mechanism of action of bapineuzumab.” The interpretation is that the treatment-related edema develops early on as large quantities of pre-existing amyloid are being mopped up by the antibody and then occurs less frequently once more amyloid has been cleared.
This mechanism would explain why ApoE4 carriers, who have higher amounts of Aβ in their brains and blood vessels, are more prone to ARIA-E. It also explains why higher doses of the drug, inducing greater Aβ clearance, would increase the risk for ARIA-E.
Not Just Bapi: ARIA Seen With Other Treatments
Scientists have suspected for some time that these side effects may be a broader phenomenon of amyloid removal from the brain (see ARF HAI story). In another poster at AAIC, Sperling described the occurrence of vascular edema and microhemorrhages in patients participating in a Phase 2 trial of the γ-secretase inhibitor BMS-708163, developed by Bristol-Myers Squibb (see Drugs in Clinical Trials). ARIA-E was found in three out of 100 patients receiving the drug, compared with zero out of 29 patients receiving placebo. All three were ApoE4 carriers. In one of the three patients, several microhemorrhages were present before treatment started and increased in number and severity during the trial. “This is just three patients, so I don’t want to speculate that this is the same mechanism,” said Sperling. “All I can say is that, at this stage, it looks very similar to the ARIA-E that we detected in patients receiving immunotherapy. No one expected to see this in this γ-secretase inhibitor trial.”
The BMS team itself presented a poster on cerebral microbleeds in a Phase 2 trial of BMS-708163 in people with mild to moderate AD. Led by Howard Feldman, these authors reported that baseline and six-month scans in 175 trial participants revealed the presence of such microbleeds in 26 percent of people, plus, at six months, a 10 percent rate of incident microbleeds in the placebo group, and 13 to 19 percent in the treatment groups. People who had one or more microbleeds at baseline were more likely to have another during the trial, though that did not affect people’s clinical outcome, the scientists reported. This study did not use the new ARIA terminology presented at AAIC.
Preliminary results from ongoing Phase 3 trials of solanezumab, Lilly’s Aβ antibody (see ARF related news story and ARF ICAD story), indicate that “less than 0.5 percent of our patients have vasogenic edema,” said Eric Siemers at Lilly. “We never had seen it in the Phase 1 and 2 studies.” As these trials are ongoing and blinded, Siemers does not yet know which patients with the vasogenic edema belong to the placebo or treatment groups.
Presentations on clinical trials data of intravenous immunoglobulin (IVIG), an old treatment by Baxter Bioscience now being tested for possible use in AD (see ARF related news story and ARF ICAD story), did not provide any information regarding the occurrence of ARIA in IVIG-treated AD patients. However, Lakshman Puli of the University of Eastern Finland, Kuopio, presented data from a Baxter-funded study showing that IVIG does not cause microhemorrhages in a mouse model of AD.
These studies are helping elucidate the mechanisms underlying vascular side effects seen in AD patients, but getting more definitive answers about their clinical impact will have to wait until results of the Phase 3 trials of amyloid-lowering therapies are available. Key questions for bapineuzumab are whether the drug is effective in treating AD and whether its benefits will outweigh side effects. “An extension to these questions is whether any beneficial effect will be greater in patients who have vasogenic edema,” said Gilman. “One possibility is that vasogenic edema is an indication that the drug is removing amyloid-β. Whether or not vasogenic edema has to occur for the antibody to work remains to be determined.”
Another important question is to what extent ARIA-E occurs as part of the natural history of AD. One of the presentations at AAIC by Christopher Carlson from Eli Lilly looked at the prevalence of vasogenic edema and microhemorrhages at baseline—in other words, before patients receive any treatment—in two ongoing multicenter, randomized, double-blind, placebo-controlled Phase 3 trials of solanezumab. The study identified two cases of ARIA-E in baseline MRI scans from 2,134 patients. “It seems that ARIA-E rarely occurs spontaneously, and it is probably more likely to occur in people with high vascular amyloid burden,” said Sperling.
New ARIA Guidelines
The talks and posters at AAIC came on the heels of new recommendations by the Alzheimer’s Association working group, published online July 12 in Alzheimer’s & Dementia (Sperling et al., 2011). The recommendations ease some of the safety restrictions the U.S. Food and Drug Administration (FDA) had previously put in place. “I think that when the FDA first identified the problem, they were correct in establishing strict criteria,” said Andy Satlin at Eisai Inc. in Woodcliff Lake, New Jersey. Satlin has no role in the bapineuzumab trials, but oversees Eisai’s/Bioarctic’s clinical trials for an antibody against protofibrillar forms of Aβ (see ARF related news story). “Then, with the available data, we were able to determine more appropriate criteria that everyone has agreed to.”
The original FDA guidelines directed clinical trial sponsors to exclude participants with more than two existing brain microhemorrhages from studies. But after reviewing all publicly available data, the working group proposed that participants with up to four pre-existing microhemorrhages (or ARIA-H) could enroll in clinical trials. Contrary to what was suggested by a number of news stories that appeared immediately after the working group’s publication, the new guidelines do not change the FDA’s requirements for monitoring patients for ARIA-E. Companies testing amyloid-reducing compounds need to perform frequent MRI tests on patients, typically every three months. Any patient who develops ARIA-E during the trial has to be taken off the medicine until those complications clear, and then treatment can resume. Anyone developing ARIA-H during the trial should continue to receive treatment, provided that these abnormalities do not worsen symptoms. “Microhemorrhages don’t disappear on subsequent MRIs as the edema does, and can be difficult to reliably track over time,” said Sperling, lead author on the working group’s report. “We don’t yet know what ARIA-H occurring in trials means for long-term outcome in AD patients.”—Laura Bonetta.
References
Therapeutics Citations
News Citations
- PIB-PET Biomarker Study Confirms Bapineuzumab Lowers Amyloid
- Paper Alert-cum-SfN: Bapineuzumab Published, More AN1792 Presented
- Chicago: Bapineuzumab’s Phase 2—Was the Data Better Than the Spin?
- Barcelona: Allosteric γ Modulation Moves Toward Clinic
- Sink or Swim?—New Take on Aβ Antibody’s Modus Operandi
- Chicago: Lilly’s Antibody Appears to Do No Harm, But Will It Help?
- Toronto: In Small Trial, IVIg Slows Brain Shrinkage
- Chicago: More Phase 2 News—PBT2 and IVIg
- Barcelona: Antibody to Sweep Up Aβ Protofibrils in Human Brain
Paper Citations
- Rinne JO, Brooks DJ, Rossor MN, Fox NC, Bullock R, Klunk WE, Mathis CA, Blennow K, Barakos J, Okello AA, Rodriguez Martinez de Liano S, Liu E, Koller M, Gregg KM, Schenk D, Black R, Grundman M. 11C-PiB PET assessment of change in fibrillar amyloid-beta load in patients with Alzheimer's disease treated with bapineuzumab: a phase 2, double-blind, placebo-controlled, ascending-dose study. Lancet Neurol. 2010 Apr;9(4):363-72. Epub 2010 Feb 26 PubMed.
- Salloway S, Sperling R, Gilman S, Fox NC, Blennow K, Raskind M, Sabbagh M, Honig LS, Doody R, van Dyck CH, Mulnard R, Barakos J, Gregg KM, Liu E, Lieberburg I, Schenk D, Black R, Grundman M, . A phase 2 multiple ascending dose trial of bapineuzumab in mild to moderate Alzheimer disease. Neurology. 2009 Dec 15;73(24):2061-70. PubMed.
- Sperling RA, Jack CR, Black SE, Frosch MP, Greenberg SM, Hyman BT, Scheltens P, Carrillo MC, Thies W, Bednar MM, Black RS, Brashear HR, Grundman M, Siemers ER, Feldman HH, Schindler RJ. Amyloid-related imaging abnormalities in amyloid-modifying therapeutic trials: recommendations from the Alzheimer's Association Research Roundtable Workgroup. Alzheimers Dement. 2011 Jul;7(4):367-85. PubMed.
Other Citations
Further Reading
No Available Further Reading
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.