CONFERENCE COVERAGE SERIES
International Conference on Alzheimer's & Parkinson's Diseases 2013
Florence, Italy
06 – 10 March 2013
CONFERENCE COVERAGE SERIES
Florence, Italy
06 – 10 March 2013
Read a PDF of the entire series.
More and more researchers believe that a neurodegenerative condition’s slow, inexorable takeover of a person’s brain might be driven by pathogenic, misfolded proteins that spread through interconnected neural networks and corrupt normal proteins along the way. It’s a wild idea that grew out of the prion field but is gaining acceptance. However, many basic questions remain, such as how these proteins enter and exit cells, and how they seed new aggregates. At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy, a talk describing a means by which tau and α-synuclein can penetrate cells generated considerable buzz, not least because it points to new therapeutic targets for slowing disease progression. Other presentations focused on protein egress and toxicity. Several speakers emphasized that wild-type proteins migrate as well or even better than mutant forms, making this idea more plausible as a mechanism for sporadic disease.
Following an original line of research on Aβ, studies in recent years have shown that injecting a small amount of aggregated tau into mouse brain kicks off pathology at the injection site, which then migrates through connected brain regions (see ARF related news story; ARF news story). The same process occurs with Aβ (see ARF related news story) and α-synuclein (see ARF related news story), and it apparently involves transmission from cell to cell. Marc Diamond at Washington University in St. Louis, Missouri, previously reported that extracellular tau aggregates can enter cultured cells and seed fibril formation (see ARF related news story; ARF news story), but it is unclear how this works.
How Bad Proteins Get In
In an enthusiastically received talk at AD/PD, Diamond proposed a modus operandi for tau entry. He found that cultured primary neurons swallow tau aggregates along with extracellular fluid in a form of endocytosis known as macropinocytosis, in agreement with prior reports in the literature. In electron micrographs, Diamond saw wild-type human tau fibrils “sticking like Velcro” to cell membranes before being engulfed in vesicles. Some peptides are known to trigger endocytosis by binding to heparin sulfate proteoglycans (HSPGs) on the cell surface. Tau contains lysine-rich motifs ideal for binding HSPGs, suggesting it might enter through this route. In immunostainings, Diamond saw tau fibrils co-localizing with HSPGs. Moreover, adding heparin, or the heparin mimetic F6, to the cell media prevented uptake of the tau fibrils, presumably by competing with HSPGs for binding sites on tau. Diamond also stopped tau’s entry by knocking down a gene necessary for HSPG production, or by adding chlorate, which prevents HSPG sulfation. Altogether, the data showed that HSPGs are necessary for cells to internalize tau. To confirm the findings in vivo, Diamond injected GFP-labeled tau fibrils into mouse cortex along with the F6 heparin mimetic and saw no fibril uptake, in contrast to injections of tau alone, which were quickly ingested.
What form and size of tau propagate best? Diamond used mostly full-length tau, although in some experiments he worked with a fragment consisting of the 4R core aggregation region. In contrast to some recent reports that tau fragments are the most transmissible form (see ARF related news story; ARF news story), Diamond saw better uptake of the full-length protein. Only aggregates the size of trimers or larger were internalized, he told Alzforum; the monomer was not.
Intriguingly, α-synuclein, the villain in Parkinson’s and related diseases, also binds HSPGs, suggesting the same mechanism might apply. Diamond reported that heparin blocks α-synuclein seeding in cultures, but has no effect on the entry of huntingtin protein, which does not bind HSPGs.
The data could open up new options for tauopathy or α-synucleinopathy treatments. Ideas include using heparin mimetics, antibodies that block uptake of pathogenic proteins, or small molecules that interfere with HSPG production. Diamond told Alzforum he will target transferases that add sugars to HSPGs. It remains to be seen if pharmaceutical companies will embrace this approach. Jean-Francois Blain at EnVivo Pharmaceuticals noted that HSPGs, being large, sticky proteins, are difficult to work with. They also typically act with co-receptors, implying that another protein could be involved in tau internalization, Blain said.
Toxic Mysteries of Tau
In addition, Diamond found that cells that took up tau seemed to acquire distinct strains of misfolded protein that varied in their toxicity, solubility, ability to seed, and aggregation pattern. For example, Diamond’s “strain 9” produces a punctate pattern of staining inside neurons and consists of mostly insoluble tau. It propagates more readily and appears more toxic than the more soluble “strain 10,” which stains as a single perinuclear inclusion. Each strain, however, was faithfully passed on to all daughter cells over months of propagation in vitro, Diamond said. Adding lysate from a stable clone to a naïve cell culture re-created the exact strain. Diamond is collaborating with Stanley Prusiner at the University of California, San Francisco, to investigate whether different strains can be linked to specific clinical phenotypes. In response to audience questions, Diamond noted that mutant forms of tau give rise to distinct strains, and that he has not yet investigated what role tau phosphorylation might play.
Voicing a common opinion, Christian Haass at Ludwig-Maximilians University, Munich, Germany, called the work “fantastic” and promising. An engaged audience bombarded Diamond with questions. For example, Charlie Glabe at the University of California, Irvine, wondered how tau might get out of macropinosomes and into the cytoplasm. Diamond said he will use caged luciferin to look for leakage of endosomal contents. Another unanswered question concerns how tau deposits affect cells and whether they cause toxicity, said Karen Ashe at the University of Minnesota, Minneapolis. Ashe noted that cognition in mice improves when monomers are lowered but tangles remain (see ARF related news story). Diamond agreed that soluble oligomers, not large deposits, probably transmit toxicity.
Other talks complemented Diamond’s findings with in-vivo data. Karen Duff at Columbia University, New York City, extended her findings from transgenic mice that express mutant human tau only in the entorhinal cortex. From there, tau pathology spreads to the hippocampus and other connected regions (see ARF related news story). By the time the mice are two years old, functional MRI reveals hypometabolism in the entorhinal cortex and hippocampus, but the animals still perform normally in the Morris water maze. Duff noted this is analogous to what is seen in people at Braak stage I or II, who have reduced brain metabolism on FDG-PET but no overt cognitive problems.
Modeling Sporadic Disease With Wild-Type Tau, α-Synuclein
Luc Buée at the University of Lille, France, wondered whether wild-type human tau migrates as readily as the mutant variety. Most studies of tau propagation have used mutant forms, but a majority of tauopathy cases are sporadic and involve only the wild-type species. First author Morvane Colin injected lentiviral vectors encoding human wild-type tau into rat hippocampus. Over five months, the protein spread to connected areas such as the limbic and olfactory regions, where it appeared inside neurons. By contrast, injections of mutant P301L tau stayed near the injection site, Colin reported. Buée speculated that the mutant protein may clump up more quickly, leaving fewer soluble species able to migrate through axons. The findings further implicate this mechanism of transmission in sporadic disease, Buée said.
Other groups are focusing on wild-type α-synuclein transmission as a model for sporadic PD. Virginia Lee at the University of Pennsylvania, Philadelphia, reviewed a model her group created by injecting wild-type human α-synuclein fibrils into the dorsal striatum of wild-type mice. Pathology spreads through the brain along anatomical connections, killing dopaminergic neurons by one month and impairing movement by six months, with cognition remaining normal (see ARF related news story). “The accumulation and spread of α-synuclein pathology reconstructs a neurodegenerative disease phenotype in healthy animals, kicked off by a single catastrophic event,” Lee said.
In the same vein, Elodie Angot, who works with Patrik Brundin at Lund University, Sweden, investigated the cell-to-cell transfer of wild-type human α-synuclein in vivo. She injected an adenovirus encoding the protein into the substantia nigra of wild-type rats. Three weeks later, all neurons in the substantia nigra expressed the protein. Then she grafted embryonic dopaminergic cells into the striatum of these rats. Two more weeks later, about a quarter of the grafted cells contained human α-synuclein, showing propagation from the substantia nigra to the striatum. The protein co-stained with an endocytosis marker, suggesting α-synuclein enters through endosomes, in agreement with Diamond’s tau data. Intriguingly, this wild-type α-synuclein spurred the aggregation of endogenous protein. Immunostaining showed the human protein forming the center of α-synuclein deposits. After treatment with proteinase K, which digests the human protein, the center of each aggregate disappeared (see Angot et al., 2012). An important caveat to the relevance of this finding for human disease is that the human protein was overexpressed.
Angot also spotted α-synuclein aggregates in oligodendrocytes. Since these cells do not express the protein, it must get in from outside. In the rat model, the aggregates, called glial cytoplasmic inclusions, appeared in immature, mature, and myelinating oligodendrocytes. Cultured oligodendrocytes readily took up monomers or small oligomers, but not lightly sonicated fibrils, which got stuck on the cell membrane, Angot reported. During the discussion, John Trojanowski at UPenn suggested that more thoroughly sonicated, broken-up fibrils might be internalized. The findings may be relevant to human disease. For example, α-synuclein deposits appear in oligodendrocytes in multiple system atrophy (MSA). However, questions remain about how well the findings will translate, scientists said. Another pathologist noted that in patients, oligodendrocyte precursor cells do not contain deposits, at odds with the in-vivo rat data.
How Bad Proteins Get Out
With all this attention paid to α-synuclein’s entry into cells, what about its exit? In Florence, Kostas Vekrellis at the Academy of Athens, Greece, addressed this issue. Although Vekrellis previously reported that α-synuclein can be secreted via exosomes (see Emmanouilidou et al., 2010), his recent studies suggest that this is not actually the primary route. In collaboration with Lawrence Rajendran at the University of Zurich, Switzerland, Vekrellis examined secretion in a neuronal cell line that overexpresses mutant α-synuclein. Vekrellis sonicated exosomes to break them open and measured their contents. He found that these vesicles contained but 1.5 percent of the total secreted α-synuclein. During the discussion, Michael Schlossmacher at Ottawa Hospital, Ontario, Canada, said that he sees similar values in his experiments. Vekrellis suggested that cells try to package α-synuclein in exosomes, but after the capacity of this pathway has been maxed out, they release free protein. He does not yet know whether free and exosome-associated α-synuclein represent the same species. For example, although exosome release is small, it could be that these vesicles carry the toxic species, he told Alzforum. Here, too, overexpression in cell cultures makes transfer of the results to the human brain difficult.
Vekrellis’ previous studies suggested that intracellular calcium regulates the release of exosomal α-synuclein. To look at free protein, Vekrellis infused depolarizing agents such as potassium chloride into the striatum of transgenic A53T α-synuclein mice by way of reverse microdialysis. Depolarization opens channels that flood cells with calcium, and, as expected, cells pumped out more free α-synuclein. Surprisingly, however, blocking potassium channels regulated by the sulfonylurea receptor 1 subunit (SUR1) cut α-synuclein secretion by half, even though this manipulation depolarizes cells and increases intracellular calcium. Likewise, activating SUR1 channels increased secretion. Vekrellis suspects the mechanism behind this may have to do with these channels affecting the burst firing of neurons, he told Alzforum. The SUR1 channel blocker glyburide is an approved medication for type 2 diabetes.
On the broader question of how these pathogenic proteins get out of neurons, a recent paper frequently came up in talks and hallway discussions. It suggests that neurons release tau quite physiologically in an activity-dependent manner that is influenced by intraneuronal calcium (Pooler et al., 2013).
These talks represent only some of the data on protein propagation featured at AD/PD 2013. We invite readers to highlight other pertinent posters and presentations through Alzforum commentary.—Madolyn Bowman Rogers.
Read a PDF of the entire series.
Call it nanonova. Okay, the analogy is far-fetched, for unlike a supernova, an exploding neuron hardly spews out tons of debris at lightning speed. But if Charlie Glabe is right, an exploding neuron does expel material that then triggers the formation of new, stable matter in its surroundings, and this is how amyloid plaques are born. This, at least, was one of the more stellar narratives to emerge from the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy. In a provocative talk that reverberated in the hallways, Glabe, at the University of California, Irvine, showed images of dying neurons, their nuclei—yes, nuclei–stuffed with amyloid. The work came out of a project to create tools to distinguish between the different flavors of the Aβ peptide, in particular to detect the small oligomeric forms that many scientists believe to be the most deadly. The data could help resurrect an old hypothesis, which holds that ruptured neurons form the core of neuritic plaques. On the broader question of how to visualize and track oligomeric forms of pathogenic proteins, numerous talks and posters at AD/PD 2013 showcased conformation-specific antibodies.
Neuritic plaque contains fibrillar amyloid (red), APP-CTF (green), and DNA (blue), suggesting its nuclear origin. Image courtesy of Ricardo Albay III
“This is a bold idea,” Gunnar Gouras at Lund University, Sweden, told Alzforum. “Glabe has been a trailblazer in this area.” Gouras pointed out that traditional ways of looking at amyloid often involve grinding up brain tissue, but antibodies such as the one Glabe made can see native protein forms in unfixed tissue, providing a glimpse into the natural pathophysiology. Other scientists expressed surprise at seeing evidence of Aβ in the neuron’s nucleus, a place the peptide is not thought to frequent.
Glabe previously generated antibodies specific for oligomeric Aβ by using a synthetic peptide, and he is developing an active vaccination strategy with this peptide as a potential therapy (see ARF related news story; ARF news story). In Florence, Glabe asked a broader question: Is the wide variation in Aβ species truly significant for the pathogenesis and heterogeneity of Alzheimer’s disease? To address this, Glabe immunized rabbits with Aβ42 and analyzed the resulting antibodies on an array dotted with 132 different forms of amyloid, including Aβ monomers, oligomers, fibrils, and islet amyloid polypeptide. He selected for further analysis 24 antibody clones that each gave different patterns of reactivity on the array. This process generated several new research tools, he noted, such as antibody M31, which stains only vascular amyloid deposits.
Do Dying Neurons Seed Plaques?
Most of his AD/PD talk, however, focused on antibody M78, which recognizes fibrillar protein forms made up of parallel β-sheets. M78 is not specific for Aβ; it also reacts with fibrillar APP and α-synuclein deposits, Glabe said. In aged human brains with some AD pathology, M78 stains nuclear and perinuclear regions of neurons, oligodendrocytes, and astrocytes in most brain regions. It seems to mark an early stage of disease, as M78-positive brains came from people who had either normal cognition or mild cognitive impairment (MCI). M78 staining goes down as more plaques deposit.
Glabe was particularly intrigued by the abnormal morphology of M78-positive neurons. Their nuclear envelope was swollen, DNA fragmented, and cell bodies filled with multivesicular bodies. Glabe used 3xTg mice to examine these changes with age. At 10 months, these animals showed the earliest M78 staining. It was perinuclear and co-localized with β-CTF immunoreactivity, implying that APP may have been cleaved by BACE1 nearby. By 12 months, M78 staining showed up in the nucleus, while β-CTF remained on the outside. By 14 months, the antibody marked the remains of a nucleus containing diffuse DNA, in the center of a neuritic plaque surrounded by ruptured membranes that stain for β-CTF. The plaque center also stained with the neuronal marker NeuN, confirming its neuronal origin. Glabe pointed out that all of these molecules maintained the same spatial relationships with each other observed two months earlier. The findings imply that amyloid accumulation lyses nuclei, Glabe said.
“Each senile plaque arises from the demise of a single neuron,” Glabe concluded. In his model, what scientists call “dystrophic neurites” are not neurites at all, but rather the remains of the exploded nuclear envelope and endoplasmic reticulum.
Glabe noted that this is not a new idea. Michael D’Andrea at The RW Johnson Pharmaceutical Research Institute, Spring House, Pennsylvania, suggested in the year 2000 that lysed neurons form amyloid plaques (see D’Andrea et al., 2001). Numerous other researchers, such as Gerd Multhaup at the Free University of Berlin, Germany, and Frank LaFerla at the University of California, Irvine, have seen intraneuronal amyloid, although controversy has erupted over whether this is actually Aβ or APP (see ARF Webinar). Reports of intranuclear Aβ, on the other hand, are quite rare, and turned heads in Florence. Glabe is careful to note that M78 is not necessarily detecting Aβ in the nucleus; it could be aggregated APP or C-terminal fragments of APP.
Numerous researchers at the conference praised the quality of the work and debated the questions it raises. “There is definitely a cell soma at the heart of plaques,” said Virgil Muresan at the University of Medicine and Dentistry of New Jersey, Newark. He pointed out that the perinuclear compartment, where Glabe first sees Aβ and β-CTF accumulation, plays an important role in APP processing, making it a logical place for deposits to start. Christian Haass at Ludwig-Maximilians University, Munich, Germany, observed that, because M78 recognizes several types of β-sheet fibrils, further work is needed to determine exactly what protein the antibody is detecting in the nucleus. He questioned whether Aβ itself could get in, given that the peptide is typically exported. Maybe it moves in after the nuclear envelope breaks down, he suggested. Gouras suggested that live imaging of cells would help nail down the progression of events, although he acknowledged that this would be technically challenging to do. Another unanswered question is how Aβ gets inside cells in the first place. It may simply accumulate in endocytic or secretory compartments where it is processed or, alternatively, cells may take up oligomeric Aβ from the outside, Glabe said.
Specific α-Synuclein Antibodies for Parkinson’s Disease Research
Meanwhile, Parkinson’s disease researchers are also developing antibodies to various toxic forms of α-synuclein, the main pathogenic protein in Parkinson’s disease. They did not deploy the antibodies to trace the provenance of Lewy bodies, but to instead characterize their potential for biomarker or therapeutic use. Therese Fagerqvist, working with Lars Lannfelt, Martin Ingelsson, and Joakim Bergström at Uppsala University, Sweden, immunized mice with α-synuclein oligomers made using an in-house process (see Näsström et al., 2011). In collaboration with BioArctic Neuroscience AB, Stockholm, Sweden, she selected for two monoclonal antibodies, mAb38F and mAb38E2, that have 150 times more affinity for α-synuclein oligomers than monomers but show no cross-reactivity with aggregates of tau, Aβ, or other synucleins. These antibodies detect pathology in A30P α-synuclein transgenic mice earlier than do commercial antibodies such as Syn-1, Fagerqvist reported. The new antibodies revealed that α-synuclein oligomer levels increase 10-fold with age in the brain and spinal cord of A30P mice. Higher oligomer levels in the endoplasmic reticulum coincide with behavioral symptoms, she noted (see Fagerqvist et al., 2013). The antibodies prevent α-synuclein oligomerization in cellular assays. When the researchers injected a related antibody, mAb47, into 14-month-old mice weekly for three and a half months, it lowered α-synuclein oligomer levels in brain extracts. The researchers are currently testing behavior.
In contrast, Gabor Kovacs at the Medical University of Vienna, Austria, described an antibody, 5G4, that he said reacts only with disease-associated forms of α-synuclein, both fibrillar and oligomeric (see Kovacs et al., 2012). 5G4 was developed in collaboration with biotech company AJ RoboScreen GmbH, Leipzig, Germany. An ongoing clinical study with 200 participants is testing its use as a biomarker of PD (see press release). In his talk, Kovacs also claimed that 5G4 detects pathology with more sensitivity than commercial antibodies. It stains deposits in the neuronal cytoplasm and processes, as well as in astrocytes. In ultrastructural studies done in collaboration with Lajos László at Eötvös University of Science, Budapest, Hungary, the researchers also see α-synuclein in endosome-like structures in neurons, supporting findings that the protein may enter cells through this route (see ARF related news story). In neurons, small α-synuclein aggregates were surrounded by clusters of mitochondria, while astrocytes contained fibrillar structures. PD patients who rapidly developed dementia had higher levels of antibody staining in limbic regions compared to those with stable cognition or slowly progressing dementia, implying that spread of α-synuclein pathology to this brain area might explain some cases of dementia associated with Lewy pathology. The antibody gives a signal even from old brain sections that have been preserved for years in formalin, suggesting it could be useful in re-evaluating archival material, Kovacs added.
Several other groups presented posters in this area. Omar El-Agnaf at United Arab Emirates University, Al Ain, has made several monoclonal antibodies specific for either oligomeric (Syn-O1, Syn-O2, Syn-O3, Syn-O4) or fibrillar (Syn-F1, Syn-F2) forms of α-synuclein, which he reported to have low affinity for monomers and do not cross-react with aggregates of tau, Aβ, other forms of amyloid, or synuclein. In previous work using a different antibody, El-Agnaf showed higher levels of α-synuclein oligomers in the cerebrospinal fluid of PD patients compared to controls, suggesting this could make a biomarker (see ARF related news story). Likewise, Harald Weninger and Markus Mandler at AFFiRiS AG, Vienna, Austria, described in a poster the development of their α-synuclein antibody, PD01. It curtails α-synuclein pathology and neurodegeneration in PD/DLB mice while improving their performance in the Morris water maze, Weninger said (see ARF related news story and Schneeberger et al., 2012). He stayed mum on the details of how the antibody was made or what form of α-synuclein it recognizes, however. The antibody crosses the blood-brain barrier and is in Phase 1 trials for PD.
The preceding studies represent only a smattering of the work in this area, all of which may provide researchers with new tools for investigating the role of specific conformations of pathogenic proteins in disease. We invite readers to add other examples through Alzforum commentary.—Madolyn Bowman Rogers.
Read a PDF of the entire series.
Pyroglutamate Aβ is a particularly toxic version of the peptide at the center of Alzheimer’s disease pathogenesis; in fact, some scientists claim it seeds the formation of plaques (for a review, see Gunn et al., 2010). It forms when a few amino acids are lopped off of Aβ’s N-terminus to expose a glutamate residue, which is then cyclized by an enzyme called glutaminyl cyclase (QC). The resulting pyroglutamate Aβ (pGlu-Aβ) is stickier than other forms of the peptide and, according to some estimates, constitutes up to half of the Aβ in AD plaques. “Pyroglutamate Aβ is a very pathogenic form of β amyloid, so it’s something you want to get rid of and prevent from occurring,” said Cynthia Lemere, Brigham and Women’s Hospital, Boston, Massachusetts. Researchers are targeting pGlu-Aβ in two ways: by generating antibodies against it and by inhibiting QC with small-molecule drugs. They presented the latest data on both strategies at the 11th Annual AD/PD 2013 Conference held 6-10 March 2013 in Florence, Italy.
Frank Weber of Probiodrug AG, based in Halle/Saale, Germany, presented initial clinical trial data on the company’s QC inhibitor, PQ912. It is the first one to be used in humans. In this single-site Phase 1 study, researchers tested the compound on 108 healthy volunteers, aged 18-50, at either a single daily oral dose of 10 to 1,200 mg or twice daily at 20 to 500 mg for 11 days. Participants took the drug either in liquid or pill form. Twenty-eight people took placebo.
In terms of pharmacokinetics, PQ912 performed quite well, Weber said. It reached a peak concentration in plasma rapidly—in two hours—and declined gradually over 12 hours. At six hours, the drug’s half-life in cerebrospinal fluid (CSF) was longer than in plasma, and CSF concentrations were about 30 percent of those in blood. In all, the drug appeared safe even at the high end of the dose range tested, and produced no serious adverse events thus far. It gave a dose-proportional response, where more inhibitor led to less QC activity measured in blood and CSF.
A critical parameter in drug development is the drug’s effective concentration that produces 50 percent of the maximum response (EC50), said Weber. In this case, researchers wanted to know how much of the inhibitor blocks half of QC’s activity. In both plasma and CSF, 11.3 nM was sufficient to knock out enzyme action halfway. The twice-daily 400 mg dose reached a 24 nM mean concentration in CSF and blocked an average of 70 percent of the enzyme’s activity. Is that enough to bring down pGlu-Aβ formation? Yes, Weber told Alzforum, because given the specific properties of the substrates Aβ3-42 and Aβ11-42, “even a small inhibition of the enzyme leads to a big increase in preventing cyclization of the Aβ peptide.” The research team tested other QC substrates and found that this inhibitor left them alone, even at the highest doses, said Weber.
Given the level of tolerability, the company is conducting a series of tests in elderly people at even higher doses to find out if they absorb or metabolize PQ912 differently. Next, they intend to test the inhibitor in AD patients and measure what it does to biomarkers of disease and cognition in this group. Weber said Probiodrug has yet to decide at what stage to treat, but that it will likely be in the early stages of AD or MCI. As plaques are known to be abundant even in early AD, will the QC inhibitor help? Yes, Weber claimed, because the compound would quell new pGlu-Aβ that forms from peptides that escape deposits.
“The compound looks exciting,” said Wiep Scheper, Academic Medical Center, University of Amsterdam, the Netherlands. “The safety and toxicology look promising, and I found the extensive dose range they tested to be impressive.” About Phase 2 trials with this drug, Scheper said, “It will be important to see which patient group they are going to test—many trials have failed because they targeted patients too late in the course of the disease.”
In response to an audience question, Weber said that PQ912 inhibits isoQC—a closely related form of the QC enzyme—as well. IsoQC cyclizes CCL2, a chemokine that attracts immune cells to atherosclerotic plaques (see Cynis et al., 2011). In previous studies, QC inhibitors have been shown to reduce such pathology. “This inhibitor could have multiple therapeutic uses,” said Lemere, “not just for AD, but for atherosclerosis and other CCL2-driven conditions.” Lemere collaborates with Probiodrug.
Other researchers are exploring ways to deploy antibodies to prevent or rid the brain of pGlu-Aβ after formation. Jeffrey Frost in Lemere’s lab presented mouse behavioral data on the group’s pGlu-Aβ-specific monoclonal antibody, 07/1. These researchers recently reported that the antibody lowered general Aβ burden in APPswe/PS1 ΔE9 mice in both prevention and therapeutic pilot studies (see ARF related news story and Frost et al., 2012). Now they find that the same antibody benefits cognition.
Starting when the mice were about six months old, just about the time they begin to deposit Aβ plaques, Frost injected groups of 11-12 male transgenic mice with either 150 or 500 micrograms of 07/1 or 200 micrograms of a control Aβ antibody, 3A1, weekly for seven months. Twelve transgenic and 12 wild-type mice injected with saline served as controls. At 12 months, the 07/1 high-dose group outperformed the saline-treated transgenics on the water T maze (see Locchi et al., 2007), nearly matching wild-type. Those receiving the 07/1 low dose showed a trend for improvement in contextual fear conditioning compared to the PBS-treated transgenic mice, and performed almost as well as wild-type mice. In general, 07/1-treated mice performed better than 3A1 treated mice. At 13 months, the mice were sacrificed. Relative to transgenic controls, 07/1-treated mice had 20-25 percent less plaque burden in the frontal cortex and 35-45 percent less in the hippocampus. Microgliosis and astrocytosis had come down as well. Based on these results, Frost told the audience that “monoclonal antibodies may trap this toxic form of Aβ, sparing mice from neurodegeneration and cognitive dysfunction.”
Frost did not find pGlu-Aβ in the periphery with use of the antibody, arguing against a peripheral sink effect. One listener asked what mechanism the researchers proposed instead. The antibody may work through microglial phagocytosis, responded Lemere, who chaired the session. Another said that the antibody’s behavioral effects seemed small. Lemere responded that the changes were statistically significant, and that mutant mice sometimes performed up to wild-type standards. Scheper pointed out that pGlu-Aβ levels are lower in mice than in humans, and considers it notable that antibodies against the rarer peptide can remove a significant portion of Aβ deposits. “That points to an important role for pGlu-Aβ in formation of the pathology,” she told Alzforum.
Exactly how pGlu-Aβ relates to plaques—whether it seeds them or forms as a modification on existing plaques later—remains controversial among scientists.
Scheper presented her own recently published work on a potential trigger and subsequent consequences of rising QC levels in aging or AD brains, which she and other groups have shown. In neuroblastoma cells, falling calcium levels in the endoplasmic reticulum—a cellular change that has been reported in human aging—led to elevated mRNA and activity of QC (see De Kimpe et al., 2012). It also resisted degradation in vitro while disrupting the lysosomal membrane in neuroblastoma cells (see De Kimpe et al., 2012). This likely allows lysosomal enzymes to leak into the cytoplasm, Scheper said. In postmortem brain tissue, she found pGlu-Aβ had built up in lysosomes of neurons and glial cells. Such cell biological havoc reinforces the rationale of targeting pGlu-Aβ therapeutically, she told Alzforum.
In Florence, another group presented human data. Milos Ikonomovic, University of Pittsburgh, Pennsylvania, showed that fibrillar pGlu-Aβ correlates with severity of disease and poorer cognition. Ikonomovic analyzed tissue samples from the posterior cingulate cortex of 64 participants in the Religious Orders Study who had had either early AD, mild cognitive impairment (MCI), or had been cognitively normal when they died. In all samples, levels of soluble pGlu-Aβ were about 100-fold lower than soluble Aβ42, probably because the pyroglutamate form aggregates more rapidly, Ikonomovic said. Soluble pGlu-Aβ levels are likely too low to be clinically relevant, and showed no association with progression of disease or cognitive impairment, he said. By contrast, fibrillar pGlu-Aβ, and both soluble and insoluble Aβ42, were more abundant in early AD than in MCI or cognitively normal samples. Higher levels of all three forms correlated with more advanced Braak stages, CERAD pathology scores, and worse performance on tests of cognition and episodic memory. Ikonomovic concurred that pGlu-Aβ may be a viable target for treatment, and said it may have potential diagnostic value.—Gwyneth Dickey Zakaib, with reporting by Madolyn Bowman Rogers
No Available Comments
Read a PDF of the entire series.
As magnetic resonance imaging (MRI) resolution improves and more animal models become available, scientists are stepping up their use of mouse MRI. But wait a second—the field has already developed MRI methods for humans; why advance them in mice? At the 11th International AD/PD Conference held 6-10 March 2013 in Florence, Italy, presenters made their case. Longitudinal MRI studies in mice, which aim to track disease progression over time, will help translate findings from animal to human, they explained.
“Not only will we understand our models much better, but we will be able to better see the effects of therapies in mice,” said Nick Fox, University College London, U.K. For example, in immunotherapy, the MRI response of Alzheimer’s patients—shrinkage in people who appear to respond to the vaccine or antibody—has surprised researchers, and having this outcome measure available in mice would enable a more thorough understanding of it.
Researchers use MRI techniques in humans to image brain characteristics such as anatomy in structural MRI, oxygen consumption in functional MRI (fMRI), and white matter integrity in diffusion tensor imaging (DTI). All of these approaches are currently used to follow how disease worsens in people. The problem is that scientists use different methods to understand the course of disease in mice. For instance, they may sacrifice animals at multiple time intervals to measure amyloid or tau pathology. Such methods are variable, costly, and require large numbers of mice. By developing non-invasive MRI technology for mice, scientists can measure disease in the same mouse, compare results between animals and people, and use fewer animals in research. “Instead of getting just a snapshot of a process, you can follow it over the whole disease course,” said Jan Klohs, University of Zurich, Switzerland.
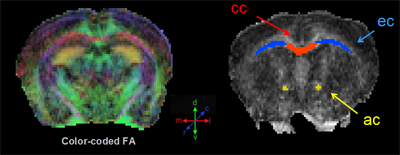
Color-coded fractional anisotropy map (left); manually drawn regions of interest (right) include the corpus callosum, external capsule, anterior commissure. Image courtesy of Moira Marizzoni
Which aspects of disease are amenable to modeling in this way? Gianluigi Forloni, Mario Negri Institute of Pharmacological Research, Milan, Italy, presented data that highlight atrophy of some brain regions in Alzheimer’s disease animal models. He and colleagues compared structural MRI results between wild-type mice and single (PDAPP), double (APP/PS1), or triple (TauPS2APP; Grueninger et al., 2010) transgenics. Over time, the volume of the putamen shrank in all three mutants relative to controls, suggesting it could be a good disease marker in these mice. “That was a surprise because we expected to see an effect in the cortex and in the hippocampus, where there is more accumulation of β amyloid,” Forloni told Alzforum.
Why the putamen, wondered audience members? This is a brain area affected in Parkinson’s disease. The authors are not sure, but researchers see abundant striatal plaque deposition in humans with familial AD, which these mice model, said Forloni. The putamen is part of the striatum. In fact, amyloid PET retention in familial AD frequently begins in the striatum (ARF related news story). Forloni’s group next plans to investigate whether the mice’s striatal atrophy is due to cell loss. In the future, they may use this marker to see how drug treatments affect this volumetric decline.
Mice may also model white matter damage, said Moira Marizzoni, of IRCCS Fatebenefratelli in Brescia, Italy. She performed a longitudinal DTI study on single (PDAPP), double (TASTPM; Howlett et al., 2004), and triple (TauPS2APP) transgenic mice to find biomarker differences from wild-type. Several markers of white matter distinguished double transgenic mice from controls. Marizzoni found a 10-15 percent change in fractional anisotropy, radial diffusivity, and axonal diffusivity mainly in the corpus callosum and anterior commissure, and less in the cerebral peduncle. Human AD patients have similar white matter changes in the corpus callosum (see Acosta-Cabronero et al., 2012). These results suggest that, once validated, these biomarkers could be promising markers of disease progression and drug effects on white matter damage in mice, Marizzoni believes. However, the single and triple transgenic mice did not show these white matter changes.
Mice can model functional connectivity, suggested Joanes Grandjean, ETH Zürich, Switzerland, in a poster presentation. Using resting state fMRI in the ArcAβ mouse model, Grandjean found that the sensory and motor cortices in the transgenics were less correlated with their respective contralateral sides than in wild-type. These differences showed up when mice were just five months old, two months before plaque deposited. Because of concerns that the anesthesia required to keep mice still in the scanner could interfere with functional connectively, researchers are moving toward doing such studies in more alert animals, he told Alzforum.
Another advantage of developing these MRI techniques in mice is that single pathologies can be observed and specifically targeted for intervention, without the comorbidities and mixed forms of dementia that complicate the picture in humans, Grandjean said. “This allows for more homogeneous groups and requires a smaller sample size than needed for human studies.”
Technological improvements have been a boon to mouse MRI, said Manfred Windisch of the CRO QPS, formerly JSW, in Graz, Austria. Stronger magnets grant higher resolution to visualize minute structures in the mouse brain, and special hardware and software reduce the signal-to-noise ratio. “You can collect a vast number of data in a single mouse, and get broader information about a drug’s effect,” he told Alzforum. Treatment studies in Alzheimer’s would benefit particularly from amyloid PET in mice, but despite one reported technical improvement (see ARF related news story), mouse amyloid PET is still not a viable tool.
On MRI, too, the researchers will hopefully go further with their data, said Menahem Segal, Weizmann Institute of Science, Rehovot, Israel. Segal chaired a session on this topic, and said he was disappointed not to see the reported MRI changes correlated with cognitive deficits, which are readily obtainable with mice. Such data would have strengthened the results, he told Alzforum.—Gwyneth Dickey Zakaib.
No Available Comments
Read a PDF of the entire series.
With variants that boost risk for both familial and sporadic Parkinson’s disease, the LRRK2 gene beckons as a prime target for therapy development. But there’s a hitch: No one knows yet how leucine-rich repeat kinase 2, as the gene is formally called, promotes disease. Basic research has rustled up a plethora of functions (see ARF related news story; ARF news story), but thus far failed to pin down a clear link to disease. A few ideas seem to be taking flight, however. At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy, researchers intrigued the crowd with compelling evidence for an inflammatory role, strengthened evidence that LRRK2 affects endocytosis and autophagy, and pointed to kinase activity as a plausible target for intervention.
Something New and Inflammatory
Most research on LRRK2 has focused on what it does in neurons, but Michael Schlossmacher at Ottawa Hospital Research Institute, Ontario, Canada, turned his eyes to the immune system. He pointed out that variants of the LRRK2 gene also associate with the inflammatory bowel disease Crohn’s and with leprosy, which is caused by susceptibility to the bacteria Mycobacterium leprae and lepromatosis. This suggested to Schlossmacher that the gene might function in inflammation. His group found rampant LRRK2 expression in circulating monocytes, which become macrophages in tissue, in neutrophils, B cells, and T cells (see Hakimi et al., 2011). Moreover, LRRK2 expression in these cells takes off after exposure to bacterial or viral particles, or when the cells are stimulated by the cytokine interferon, which rallies the immune system to fight infection. LRRK2 expression also spikes in white blood cells seen in capillaries of brains that are infected with rabies or HIV, or are afflicted with various forms of PD, Schlossmacher reported. Likewise, the protein burgeons in other types of inflamed tissue, such as leprosy skin biopsies, lymph nodes, and spleen.
How might LRRK2 promote PD from inside immune cells? Not by regulating cytokine release, as cells with mutant LRRK2 performed as well as wild-type cells in this regard, Schlossmacher said. He speculated that the gene could be involved in phagocytosis, the process by which macrophages gobble up harmful substances. He plans to look at this next. A phagocytic role would dovetail with findings that LRRK2 helps regulate neuronal endocytosis, a similar process, he said. Overall, the data suggest that pathogenic LRRK2 variants might increase a person’s susceptibility to Parkinson’s by weakening the immune system. The penetrance of dominantly inherited PD-associated LRRK2 alleles is only about 25 percent, and it varies with ethnicity and geography, Schlossmacher noted. This suggests that an environmental trigger, such as an infection, is needed for LRRK2 to cause PD (see Kitada et al., 2012).
Bart De Strooper at KU Leuven, Belgium, told Alzforum he was impressed by the data showing immune cells loaded with LRRK2, and said that these cells deserve further study. For his part, De Strooper recently reported that in flies, LRRK2 phosphorylates endophilin A, a protein involved in synaptic vesicle endocytosis (see ARF related news story). Endocytosis is disrupted both when there is too much and too little LRRK2 activity. This means that researchers targeting hyperactive LRRK2 with inhibitors should be careful not to overdo it, he suggested. In Florence, De Strooper extended these results to mice. He is collaborating with researchers at Janssen Pharmaceuticals, Beerse, Belgium, who showed data confirming his endocytosis findings in a poster presentation.
Something Old—Role in Autophagy
Several Parkinson’s risk genes, including LRRK2, have been previously linked to autophagy, a waste disposal system inside cells (see ARF related news story). The autophagy narrative has become a hot topic in PD research, since a blockage in this process may help explain why cells accumulate α-synuclein deposits. In Florence, researchers elaborated on the story. For example, Schlossmacher reported that levels of a key autophagy marker drop in immune cells containing mutant LRRK2, suggesting this pathway is impaired.
Ben Wolozin at Boston University, Massachusetts, found that numerous autophagy genes are co-regulated with LRRK2 (see Ferree et al., 2012). In a worm model, these genes interacted with pathogenic LRRK2 variants to damage dopaminergic neurons. In mammalian cells, Wolozin showed that one such gene, histone deacetylase 6 (HDAC6), directly binds LRRK2 and mediates the ability of the G2019S LRRK2 mutant to dial down autophagy. G2019S is the most common PD-associated variant, and causes hyperactivation of the kinase domain.
Wolozin then wove α-synuclein and the effects of aging into this picture. He crossed worms carrying either wild-type or G2019S LRRK2 to animals marked with a reporter molecule for autophagic flux. In young worms, the presence of wild-type LRRK2 pumped up autophagy, while the mutant form had no effect. However, in middle-aged and older worms, mutant LRRK2 dampened autophagy. Then Wolozin added human α-synuclein to the animals, which lack an endogenous version of this protein. Again, in young worms, the protein caused no problems, regardless of which LRRK2 variant they expressed. In old worms it was a different story. Animals expressing α-synuclein with either form of LRRK2 lost dopaminergic neurons. The G2019S variant led to slightly more cell death and much less autophagic flux than did the wild-type version, but both were harmful. Wolozin and colleagues previously found that LRRK2 jacks up α-synuclein expression (see Carballo-Carbajal et al., 2010). With age, this may lead to too much α-synuclein, resulting in aggregation and toxicity, he speculated. It remains to be seen whether these findings will translate to people, though LRRK2 has previously been found to exacerbate α-synuclein aggregation in transgenic mice (see ARF related news story). The finding is controversial, since two other mouse studies did not turn up synergy between these proteins (see Daher et al., 2012; Herzig et al., 2012).
What About the GTPase?
Besides its kinase, LRRK2 contains a second functional domain, a GTPase. Darren Moore at the Swiss Federal Institute of Technology in Lausanne (EPFL) pointed out that some PD-associated LRRK2 variants lower activity of this enzyme, suggesting this domain could be involved in disease. In Florence, Moore described a yeast screen he used to find genes that affected toxicity due to LRRK2 overexpression. Out of nine hits, only one had a human orthologue. This turned out to be ADP-ribosylation factor GTPase-activating protein 1 (ArfGAP1). LRRK2 physically interacts with ArfGAP1, and the two proteins are found together on Golgi membranes, in synaptosomes, and in the cytoplasm, Moore reported. Not only does ArfGAP1 activate LRRK2’s GTPase function, but LRRK2 also phosphorylates ArfGAP1, suggesting the two proteins may regulate each other. Silencing ArfGAP1 rescues a LRRK2-mediated shortening of neurites in primary cortical neurons. This shows that ArfGAP1 is required for LRRK2-induced toxicity, Moore concluded (see Stafa et al., 2012). He is looking for the mechanism now, and will also see if these cell culture results hold in vivo.
Moore also investigated how the protein’s kinase and GTPase activities might interact. He made synthetic mutations in LRRK2’s GTPase domain, and found that they diminished the protein’s kinase activity (see Biosa et al., 2013). To look in vivo, he injected virally encoded G2019S LRRK2 into rat striatum. About one-third of nigral neurons took up the gene, leading to the loss of dopaminergic neurons in the substantia nigra, together with axonal degeneration and ubiquitin pathology in the striatum (see Dusonchet et al., 2011). Injecting a kinase-dead version of G2019S LRRK2, or one with an activated GTPase, on the other hand, produced much less striatal pathology. Both of these approaches might be useful for tackling the G2019S mutation, he suggested.
Will one therapy work for all LRRK2 mutations, or will treatments need to be targeted to the specific variant? Many LRRK2 mutations do not affect kinase activity, and might require different strategies, such as hitting downstream targets of the protein, Moore predicted.
Keep It Simple: It’s a Kinase
Meanwhile, pharmaceutical companies are encouraged by the fact that the most common pathogenic mutation of LRRK2 leads to a hyperactive kinase. Drug developers have extensive experience in designing kinase inhibitors. Strengthening the case for this approach, biochemical and functional studies show that too much LRRK2 kinase activity poisons neurons (see Greggio et al., 2006; Lee et al., 2010; Ramsden et al., 2011). Moreover, the mechanism could extend to non-genetic forms of the disease. Warren Hirst at Pfizer, Cambridge, Massachusetts, pointed out that LRRK2 protein levels are elevated in sporadic PD (see Cho et al., 2013). This finding suggests that overactivation of LRRK2 could be a common feature in PD, Hirst said. He is looking now to see if kinase activity is, in fact, up in sporadic brains. This will be a key issue for pharmaceutical companies, as it would greatly increase the number of people who might take a kinase inhibitor drug.
The Pfizer program aims to develop selective, brain-penetrant kinase inhibitors for LRRK2, Hirst said. One challenge is that few good tools exist for studying LRRK2. For example, researchers lack a validated animal model. Hirst noted that the rat model described by Moore looks promising. Scientists are also hampered by the lack of a validated physiological target for LRRK2, which would be helpful for measuring whether a drug is having the desired effect. Another pressing need is for pharmacological inhibitors and probes for LRRK2. Hirst described an inhibitor Pfizer has developed as a research tool. Called LRRK2-IN-1, it inhibits the kinase with 20 nM potency, but does not cross the blood-brain barrier. Pfizer has also designed a radioligand that binds LRRK2, which is enabling them to perform tissue binding studies in transgenic mouse brain and kidney. These kinds of studies provide a knowledge base for developing drugs, Hirst said. In addition, Pfizer is now testing an inhibitor that gets into mouse brain when given at 10 mg/kg. Hirst noted that LRRK2 inhibition results in conformational changes that can be detected with antibodies (see Gillardon et al., 2013; Sheng et al., 2012), which will provide another way to measure the effectiveness of inhibitors.
Only time will tell whether this pharmaceutical approach will pan out. Hirst told Alzforum that the immunological data shown by Schlossmacher looked intriguing, as they link the LRRK2 mutation to other diseases. That may provide an alternative route to the clinic, Hirst suggested.—Madolyn Bowman Rogers.
No Available Comments
Read a PDF of the entire series.
The 11th Annual AD/PD 2013 Conference held 6-10 March 2013 in Florence, Italy, featured a session sponsored by the biotech company Io Therapeutics in Santa Ana, California, at which researchers connected with the company and at an academic center proposed that a cancer drug newly proposed for neurodegenerative diseases seems to do it all—rejuvenate dopaminergic cells; break up α-synuclein, Aβ, and tau aggregates; and quell neuroinflammation. The drug, IRX4204, is being tested in a small Phase 2 trial for treatment of prostate cancer.

IRX4204 disaggregates paired helical filaments isolated from the brains of AD patients. Image courtesy of Jun Wang
“We can protect dopaminergic neurons, give them trophic support for survival, and at the same time, interfere with some of the mechanisms that form the basis for degeneration in multiple diseases,” said Giulio Pasinetti, Mount Sinai Hospital, New York, who chaired the session.
What is this multitasking drug? Martin Sanders, who is the CEO of Io Therapeutics, Inc., presented IRX4204 characteristics gleaned from its development in cancer. The compound works as an agonist for retinoid X receptors (RXRs), which are in the cell nucleus and bind either to each other to form homodimers, or to different nuclear receptors to form heterodimers. Upon activation by a ligand—either an endogenous one or a drug—these receptor complexes regulate gene transcription. Many pharma companies have been trying to develop RXR agonists for Alzheimer’s disease but have dropped them because of liver and other toxicities. Sanders emphasized that IRX4204 is extremely selective for RXR over retinoic acid receptors (RAR), claiming that it activates RXR homodimers maximally at concentrations of just 1 nanomolar and RAR homodimers at concentrations in the micromolar range. At low doses, “IRX4204 specifically activates RXR, which means the drug will not have clinically adverse events related to RAR activation,” Sanders said.
What tipped off the researchers that their drug could straddle cancer and neurodegeneration? Elizabeth Nowak, a postdoctoral fellow in the laboratory of Io co-founder and scientific advisor Randy Noelle at the Geisel School of Medicine at Dartmouth University, Lebanon, New Hampshire, told the audience that IRX4204 affects neuroinflammation and autoimmunity. In a B6 mouse strain of experimental autoimmune encephalomyelitis, a commonly used model for multiple sclerosis, treatment with IRX4204 reduced the number of myeloid dendritic cells and CD4+ T cells that crossed the blood-brain barrier to enter the central nervous system. These cells demyelinate neurons, leading to disease, but in treated mice disease was delayed and less severe than vehicle-treated controls, Nowak said. She used six to eight mice per group.
Treatment with IRX4204 was associated with lower IL-17A production by CD4+ T cells in the periphery, Nowak said. This suggests that the Th17 lineage, a population of CD4+ T cells that releases this cytokine and is considered pathogenic in this and other models of autoimmunity, was impaired. Sanders and Rosh Chandraratna, who, according to Io Therapeutics, took a series of retinoid and rexinoid compounds from the larger company Allergan to co-found the biotech company, reasoned that the drug might treat neuroinflammatory and autoimmune processes in Alzheimer’s and Parkinson’s.
Sanders and colleagues found that IRX4204 does not transactivate RXR/PPARγ or LXR heterodimers. Rather, it transactivates the RXR/Nurr1 heterodimer, which is known to promote expression of genes that play a role in dopamine synthesis and the maintenance of dopamine neurons (see Perlmann and Wallén-Mackenzie, 2004). That suggested a possible benefit for Parkinson’s disease (PD).
In a widely noted recent study by Gary Landreth, Case Western Reserve University, Cleveland, Ohio, and colleagues, a mixed RXR/RAR agonist called bexarotene reduced Aβ plaque load in APP/PS1 mice by way of ApoE-mediated clearance (see ARF related news story on Cramer et al., 2012). Reading it, Sanders and colleagues considered that IRX4204 might reduce AD plaque without side effects associated with RAR activation. To explore a possible AD/PD link, Io collaborated with Pasinetti, who tested whether IRX4204 affects expression of dopamine-related genes and cell death. Pasinetti incubated primary mesencephalic neuron cultures from mice with 1 nanomolar of the drug. These cells normally degenerate in serum-free culture, but treated cells lived longer and expressed more dopamine transporter and DOPA decarboxylase genes than untreated cells, Pasinetti told the audience. These results suggest treatment may help protect dopaminergic neurons, he said.
Other experiments suggested that IRX4204 disrupts aggregation of α-synuclein in vitro and improves behavior in mice. The drug prevented dimers and trimers of the protein from forming in photo-induced crosslinking tests, reported Pasinetti. In behavioral tests, rats that had a one-sided lesion of the nigrostriatal pathway were tube fed for three weeks with 10 mg/kg/day of the drug. They performed better on the cylinder test than untreated controls.
Jun Wang, a collaborator of Pasinetti’s at Mt. Sinai Hospital, tested the drug’s effect on Aβ42 oligomerization by using nuclear magnetic resonance spectroscopy to see whether the drug interacts with the peptide. When Aβ and IRX4204 were combined in equal concentrations, a small cluster of Aβ’s atoms changed position, suggesting to Wang that IRX4204 binds to Aβ42 and changes its conformation. In in-vitro crosslinking experiments, equal molar concentrations of IRX4204 reduced the concentration of Aβ40 and Aβ42 dimers, trimers, and tetramers, supporting the idea that the interaction between the drug and Aβ can interfere with the formation of Aβ oligomers, Wang said.
In TgCRND8 mice, an aggressive amyloid deposition model, six weeks of daily IRX4204 oral treatment of 10 mg/kg/day resulted in 4-8 nanomolar accumulations in the brain after 10 days. Treated TgCRND8 mice performed better on the Morris water maze test and tests of contextual memory than vehicle-treated transgenics. These mice demonstrated no side effects, Wang said. On brain slices, treated mice had fewer hippocampal and cortex plaques than did controls.
Wang looked for drug effects on long-term potentiation in brain slices of 18-month-old Tg2576 mice, which are known for their LTP deficit. Incubating with 300 nM of IRX4204 for one hour restored LTP almost to wild-type levels. What pathways could lead to the improvement? In mouse N2a cells, scientists found that treatment activated the CREB pathway necessary for LTP. “We think IRX4204 can prevent oligomerization, activate CREB signaling, lead to improved synaptic plasticity, and contribute to learning and memory in AD,” Wang said.
Not only that, but IRX4204 may also prevent and break up tau aggregates, Wang claimed. As in the Aβ crosslinking studies, IRX4204 prevented dimers, trimers, and tetramers of tau from forming. Wang transfected SH-SY5Y cells and HEK293 cells with GFP-labeled tau and induced its aggregation by adding paired helical filaments isolated from human brain. If she treated these cells with 1 nanomolar of IRX4204, less insoluble tau formed. In vitro, Wang said that IRX4204 unraveled tau β-sheets from human brain-derived paired helical filaments (PHFs) in a dose-dependent manner. At a concentration of 1 micromolar, 90 percent of the PHFs lost filament structure, while 10 nanomolar of IRX4204 unraveled half of the filaments in two hours. “To date, Aβ therapy has not been successful by itself,” Wang told Alzforum. “This drug can potentially target both pathological features of AD.”
Other scientists questioned the micromolar concentrations used to elicit the greatest tau effects, cautioning that such a high level of drug could have non-specific effects on nuclear receptors. Anti-aggregation compounds have failed in previous clinical trials. Alzhemed™, which was said to inhibit amyloid deposition by binding Aβ and holding it in soluble form, showed no efficacy in Phase 3 (see ARF related news story). Scyllo-inositol, which is reported to inhibit Aβ oligomerization, failed to improve cognition or function in patients with mild to moderate AD in Phase 2 (see ARF related news story), and at high doses showed serious adverse events in Phase 3 (see ARF related news story).
Io Therapeutics has completed a four-week Phase 1/2 study of IRX4204’s effects on solid tumors that were unresponsive to treatment. Eighteen patients with various types of cancer tolerated the drug without compound-related severe adverse events. Disease stabilized in half the patients for four months or longer, and in two patients for more than a year. Side effects included elevated triglycerides and hypothyroidism, both known and treatable consequences of RXR agonism, Sanders said. Alzheimer's researchers commented that while these side effects are acceptable when a person is critically ill with cancer, they may be a problem for aging patients who are taking a drug for years.
Does IRX4204 get into the human brain? Pasinetti did not answer this Alzforum question directly. He said scientists would aim to find out in a future trial. He spoke about the need to establish how much of the drug crosses the blood-brain barrier and engages its target in the brain, what its half-life is in the cerebrospinal fluid (CSF), and determine an effective concentration for clinical intervention. Experiments in mice indicate that the drug reaches nanomolar concentration in the CSF, Pasinetti told Alzforum.
Overall, other scientists at AD/PD expressed some skepticism about this drug, though most declined to be quoted by name. "These cancer biologists have made an interesting foray into neurobiology and have a series of interesting preliminary observations,” said Landreth. “However, they have not convinced me that the drug is absolutely specific for the RXR/Nurr1 heterodimer,” he told Alzforum. “Looking at their side effect profile, it seems some activity may be due to added interactions with RXR in combination with LXR or PPARγ."
That selectivity profile will be key to success for an RXR-modifying drug, said other experts. Some RXR agonists are known for their pleiotropic effects because RXR dimerizes with various other nuclear receptors. In addition, given that many of these compounds come from the cancer field, it remains to be seen whether they are safe enough for long trials in AD, they told Alzforum.—Gwyneth Dickey Zakaib.
No Available Comments
Read a PDF of the entire series.
Some dismiss it as a therapeutic dead end, but for others, the M1 muscarinic receptor is a target for ongoing drug development research in Alzheimer’s disease (AD). These receptors receive acetylcholine in the postsynaptic membrane, mediating cholinergic transmission. Since various subtypes of nicotinic and muscarinic receptors are present throughout the body, drugs that target all acetylcholine receptors or boost levels of the transmitter, as does donepezil, have nasty side effects, such as vomiting, nausea, and bladder infections. At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy, researchers presented the latest data in their search for compounds with fewer side effects that selectively target the M1 receptor. “This is a key pathway that has been successfully exploited for treating one aspect of Alzheimer’s disease,” said Dirk Beher. “Therefore, there’s a higher probability of success for new drugs that target it.” Beher recently left Merck Serono to start up Asceneuron, Lausanne, Switzerland.
Beher introduced a novel M1-targeting compound called ASN-51, currently under development by Asceneuron. ASN-51 is a highly M1-specific positive allosteric modulator (PAM). PAMs amplify the effect of the natural ligand, whereas NAMs weaken it. Beher and colleagues developed this compound starting from a hit from high-throughput screening. The compound binds to M1 at a spot other than where its ligand binds and potentiates the receptor response by heightening M1’s affinity for acetylcholine. Since the neurotransmitter wanes in AD, enhancing M1’s sensitivity to acetylcholine is likely a good thing, said Beher.
In cellular assays with CHO cells overexpressing all five types of muscarinic receptor and in mouse primary neuron cultures, ASN-51 was effective at nanomolar concentrations and modulated only the M1 subtype. Following one oral dose, the compound got into the brains of wild-type mice and elicited a dose-dependent rise in inositol monophosphate, which happens in response to activation of Gq/11 receptors that are coupled to M1, M3, and M5. After optimizing ASN-51, the company plans to see if it benefits preclinical cognition models such as those in the scopolamine challenge paradigm, Beher said.
Scientists in the audience raised concerns about peripheral side effects, such as gastrointestinal problems, seen with other compounds said to target the M1 receptor. That won’t be a problem, claimed Beher. According to the cellular profile of ASN-51, the drug is more selective for M1—the primary muscarinic receptor in the brain—than previous compounds, meaning it should affect peripheral muscarinic receptors less.
The fact that ASN-51 is a positive allosteric modulator bodes well for the drug’s safety. “PAMs are good compounds because they avoid the side effect of strong M1 activation; they only increase the endogenous signal,” said Christian Czech, who works at Roche in Basel, Switzerland, but did not speak on their behalf. This makes PAMs generally safer than agonists, he told Alzforum. Czech noted that Beher has not yet presented toxicity data, which will be necessary to move the compound forward.
Scientists at Heptares Therapeutics in Hertfordshire, U.K., are aiming for a similar goal. They want to find a specific agonist to bind M1’s acetylcholine site and activate the receptor even when the natural ligand fades from the AD brain. That is not an easy task, as all muscarinic receptor subtypes have a highly conserved binding site for the neurotransmitter, explained Alastair Brown. To find such a precise ligand, Heptares is using structure-based design, where researchers stabilize the receptor with one of a number of different agonists in the binding site, and then crystallize it. By reconstructing an image from the pattern of x-rays as they travel through the crystal, scientists can study the exact binding mode of each agonist and determine how it interacts with the protein. By comparing these data between receptors, they can discover what makes M1 binding unique and develop new molecules that fit those specifications.
In structure-based drug design, researchers stabilize a receptor with different compounds in the binding site to study how protein and ligand interact at the atomic level. This example depicts a noradrenaline receptor with a synthetic agonist; the M1-POM combination discussed in this story is proprietary. Image courtesy of Heptares Therapeutics
Through this method, Heptares generated (S)HTL-B, a compound they claim is highly selective over the M2 or M3 receptors. In rats, the drug improved discrimination in novel object recognition and counteracted a scopolamine-induced deficit in passive avoidance, Brown said. The compound’s benefits added to those of donepezil, which retains acetylcholine in the synapse by limiting its breakdown. The company is studying the compound’s toxicology and planning a Phase 1 trial, Brown said.
Another potential candidate for an allosteric M1 agonist is AF710B, a compound Abraham Fisher of the Israel Institute for Biological Research, Ness Ziona, presented at a conference in Zuers this past December (see ARF related news story). AF710B revs up secretion of sAPPα, anti-apoptotic BCL2 expression in mitochondria, and reduces tau hyperphosphorylation, GSK-3β activity, and the apoptotic Bax protein. Fisher hypothesizes that the drug acts on heterodimers of the M1 with the σ-1 receptor in the membrane of the ER.
At the AD/PD Conference, Fisher presented data from the first tests of AF710B efficacy in transgenic mice. In a chronic dosing study, Rodrigo Medeiros and Frank LaFerla of the University of California, Irvine, injected 10 μg/kg/day of the compound into the peritoneum of 10-month-old 3xTgAD female mice daily for two months. These mice performed better than untreated transgenics in the Morris water maze. Animals were sacrificed at 12 months, and treatment about halved soluble and insoluble Aβ40 and Aβ42. Tau phosphorylation fell by more than half while astrocytes, microglia, and markers of inflammation diminished. Mice experienced no side effects. Neither did rats at acute treatment of concentrations 50,000 times higher than the effective dose, said Fisher.
Fisher’s previous compound, AF267B (see ARF related news story), produced similar results in the same triple transgenic mouse strain. AF710B is more potent, said Fisher in his presentation.
Despite some apathy in the field for the acetylcholinesterase pathway, it is still an important target because AD patients need therapies that treat their symptoms better than does donepezil. “In the absence of a disease-modifying treatment, we can still aim to produce a compound with a higher efficacy that lasts for a longer period of time,” Czech told Alzforum. Even if a disease-modifying compound is found, it may not have immediate effects on AD symptoms. “If you could combine it with a good symptomatic treatment, the benefit for patients would be even higher,” Czech said.—Gwyneth Dickey Zakaib.
No Available Comments
Read a PDF of the entire series.
Adding insult to injury, many patients with a movement disorder get dementia, too. In fact, up to 80 percent of Parkinson’s disease (PD) patients develop cognitive deficits (see Aarsland et al., 2003). But researchers do not know what causes this mental decline. Is it α-synuclein pathology in the cerebral cortex? Aβ pathology from comorbid AD? Dying dopamine cells in the basal ganglia? Or all of the above? At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy, several groups showed how they are using imaging and biomarkers to parse the causes of dementia in Lewy body disorders. The more tools scientists have to look, the more the answer appears to be: all of the above.
The topic is picking up in the scientific literature as well. On March 25 in JAMA Neurology, scientists reported that mild cognitive impairment (MCI) in early PD predicts dementia down the road. Other researchers reported in the journal Movement Disorders that cerebrospinal fluid (CSF) Aβ in PD correlates with enlarged brain ventricles.
For clues about whether Aβ causes dementia in Lewy body disorders, John Growdon, Massachusetts General Hospital, Boston, told the audience at AD/PD 2013 that his group conducted cognitive tests and amyloid imaging with Pittsburgh Compound B (PIB) on normal aged controls and patients with PD, PDD, or DLB. PDD is Parkinson’s disease dementia, and DLB, dementia with Lewy bodies. Groups comprised 12 to 29 people. The most fibrillar amyloid occurred in the brains of people with DLB, followed by PDD and PD without dementia. In the DLB group, having more amyloid correlated with a greater clinical dementia rating and lower MMSE score. Those results suggest that in DLB, amyloid pathology is one of the abnormalities leading to dementia, Growdon said.
In this case-control, one-time comparison, amyloid pathology did not distinguish PD patients with mild cognitive impairment from those without. However, PET imaging with 18F-fluorodeoxyglucose did. (FDG) PET picks up hypometabolism. This supports the notion that both groups may have amyloid early on, but only those who also have synapse loss in the cerebral cortex have developed MCI, Growdon said.
Does having amyloid pathology when one has Parkinson’s predict that cognitive decline will follow? To find out, Growdon and colleagues conducted baseline amyloid scans in 35 cognitively normal people with PD and 11 people with PD-MCI, and administered cognitive tests every year for two and a half to five years. While baseline PIB uptake still did not distinguish between PD and PD-MCI groups, it did predict the speed of progression. Those who had PIB uptake above the median, i.e., had more amyloid in their brains, progressed faster to PD-MCI or PDD than those with uptake below the median (see Gomperts et al., 2013). “These results establish β amyloid as a risk factor for causing dementia in PD,” Growdon told listeners in Florence. “Whenever effective anti-amyloid therapies emerge from research for AD, such treatments will be immediately relevant for PD as well.”
Amyloid-β pathology is not the only contributor to cognitive impairment in people with Parkinson’s. Dopaminergic cell death spreading to areas important for learning and cognition also plays a part. Growdon used the PET radioligand altropane, which labels dopamine transporters, to find out if a loss of this synaptic signal was linked with worse cognition. In both DLB and PDD patients, a lower signal in the caudate correlated with cognitive impairment, suggesting that a loss of dopaminergic neurotransmission in that structure contributes to dementia in DLB and PDD, Growdon said.
Studies like this are important for designing clinical trials, said John Trojanowski, University of Pennsylvania, Philadelphia. “We have a spectrum of clinical manifestations of Lewy body pathology that can be PD, PDD, or things in between,” he told Alzforum. “Yet the emergence of dementia in these patients is not due just to Lewy bodies; there are other contributors, like plaques and tangles.” To target patients at risk for dementia in clinical trials, researchers need to understand which pathologies predispose to cognitive decline in PD, he said.
Can imaging aid in the differential diagnosis of these disorders? To find out, Andrew Siderowf, who left the University of Pennsylvania, Philadelphia, to work for Avid Radiopharmaceuticals, Philadelphia, imaged five PD patients, 11 people with DLB, 10 with AD, and five healthy controls with the amyloid tracer 18F-florbetapir and with AV-133, a PET ligand for vesicular monoamine transporter 2 (VMAT2). AV-133 is an investigational tracer. It images the protein that loads synaptic vesicles with monoamine neurotransmitters such as dopamine (see ARF related news story). Avid, which developed florbetapir (trade name Amyvid®), is developing AV-133 for clinical use in differential diagnosis. In this small study, patients with PD and DLB had abnormal AV-133 scans, much as might be expected with altropane, while those with AD and healthy controls had normal ones. “AV-133 imaging may be useful for ruling out AD because, in general, patients with AD have normal scans,” said Siderowf.
Most clinically diagnosed AD patients had evidence of amyloid deposition. DLB patients either had both amyloid and dopaminergic loss, or only the latter, making them a mixed group. In addition, patients with less dopamine signal in the caudate had a lower MMSE score. “That could be a biomarker signal that explains part of why patients with DLB have cognitive problems,” Siderowf told Alzforum, noting that his results fit well with Growdon’s. “The association between cognition and dopamine loss in the caudate in Lewy body disorders has not been widely reported,” he said. “It’s reassuring to know that our results are consistent.”
Amyloid and dopamine imaging in people with DLB may help shed light on the heterogeneity of this disorder, and aid prognosis and treatment in the future, he added. In this early imaging study, the participants’ diagnoses were not autopsy confirmed.
Both research groups necessarily compared apples and oranges. For AD, they had a way to visualize a defining protein pathology but not cholinergic neurotransmission, whereas in Lewy body disorders, they are able to image damage to dopaminergic neurotransmission but not the defining protein pathology. Imaging agents to visualize tau, a defining pathology in AD that is also thought to be involved in some Lewy body disorders, are only just entering human testing.
In the absence of a complete set of imaging tools, levels of cerebrospinal fluid (CSF) Aβ42 may also predict cognitive decline in PD, according to Lucia Farotti, who works with Lucilla Parnetti at the University of Perugia, Italy. Farotti monitored CSF biomarkers and cognition longitudinally in 56 patients with PD. If participants' Aβ42 exceeded 800 picograms/milliliter in the CSF at baseline, their MMSE scores stayed stable up to eight years. Those with levels under 800 pg/ml at baseline declined by an average of two points during that time. CSF Aβ42 levels predicted cognitive decline in PD patients with a sensitivity of 80 percent and a specificity of 54 percent, Farotti said. These results add to the argument that AD pathology is a predictor of cognitive impairment in PD, and could be a useful prognostic biomarker, she concluded. The data confirm previous findings by other labs of lower CSF Aβ in some people with PD, and add longitudinal observation.
Seen clinically, mild cognitive impairment, too, predicts dementia in PD. That is according to one of the first studies on the prognosis of MCI in PD, published online March 25 in JAMA Neurology. A previous report on advanced PD patients found that 62 percent of those with MCI progressed to dementia over four years, versus 20 percent who did not have MCI at baseline (see Janvin et al., 2006). This is the first study to examine the same issue in early PD, said Kenn Freddy Pedersen, Stavanger University Hospital, Norway. He and colleagues reported three-year data from the prospective Norwegian ParkWest study, which followed 182 non-demented patients with newly diagnosed PD for three years. By the third year of follow-up, 27 percent of those with MCI at baseline were diagnosed with dementia, compared to only 0.7 percent of those without. Translating to a 9 percent annual rate of progression, this estimate roughly equals that found for AD (see Petersen et al., 2009), wrote the authors.
Some participants reversed their cognitive decline. One in five people with PD-MCI at baseline tested normally at the three-year follow-up, again within limits of the 14-41 percent range reported for non-PD populations. Of those who had PD-MCI at both baseline and one year, 45 percent progressed to dementia after three years and only 9 percent reverted. That suggests repeated tests strengthen the prognostic value of MCI, wrote the study authors. With a sensitivity of 91 percent and a negative predictive value of more than 99 percent, MCI within the first year of PD diagnosis is a strong predictor for further cognitive decline, they concluded.
That many people reverting to normal cognition “suggests that PD-MCI is not a stable finding,” commented Brian Copeland and Mya Schiess of the University of Texas Medical School at Houston, in an accompanying editorial. “Any enthusiasm over the predictive value of the diagnosis must be tempered with that in mind,” they added. MCI is a clinical diagnosis. Another hint on how to sharpen such a clinical diagnosis came just this month from the same ParkWest study in Norway. In this month’s issue of Movement Disorders, Mona Beyer at Stavanger, working with imaging researchers led by Liana Apostolova at the University of California, Los Angeles, link low CSF Aβ to ventricular enlargement in the study participants. Since expanding ventricles were previously associated with disease progression in MCI, AD, and PD, CSF and MRI markers may help identify patients at risk for cognitive decline, wrote the authors.
In recent years, Tamas Revesz and colleagues at the Queen Square Brain Bank (QSBB), London, U.K., have published data about dementia in PDD gleaned from histopathological studies of postmortem human brains. In an AD/PD 2013 conference presentation reviewing these findings, Revesz highlighted a role for Aβ and tau pathology in addition to Lewy bodies. All three pathologies are present in those who died with PDD, and a combination better predicts dementia than any one of them alone. Interestingly, Aβ and α-synuclein seem to interact, where a higher level of one tends to come with more of the other. “If you have a treatment that modifies α-synuclein pathology, that one therapy may not be sufficient to treat these individuals. You may also need to target Aβ pathology,” said Revesz, echoing Growdon’s statement about the potential value of future anti-amyloid medications for patients at risk of cognitive decline in PD.
These studies are important because they generate data on larger numbers of participants that together enable scientists to understand what is causing dementia in Lewy body disorders, said Trojanowski. “We need more autopsy data with longer and better follow-up of patients within the last few months or years of life to get a more secure idea of what causes the dementia.” With such information, scientists may be able to identify who is going to get PD-related dementia before they exhibit symptoms, said Pedersen, adding, “If we could reach these patients before they become impaired, we could perhaps delay dementia.”—Gwyneth Dickey Zakaib with reporting from Madolyn Bowman Rogers
No Available Comments
Read a PDF of the entire series.
After more than a decade of seemingly pushing against a wall, BACE inhibitors for Alzheimer’s disease are finally surging forward. At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy, several pharmaceutical companies provided sunny updates on their candidate drugs, which have all shown the ability to dramatically lower Aβ in the cerebrospinal fluid (CSF) in Phase 1 trials. Most of the drugs have been tested only on healthy volunteers to date, but notably, Merck presented Phase 1 data from AD patients. Their compound was as effective in lowering Aβ in this population as it had been in the healthy controls, said Merck’s Mark Forman. The talks represented but a handful of the companies with inhibitors currently in Phase 1 or 2, which at this point all appear to look similar. In Florence, academic and pharmaceutical researchers alike expressed optimism that targeting BACE1 could be a viable therapy.
“I am impressed by how well BACE inhibition is working,” Michael Willem at Ludwig Maximilians University, Munich, Germany, told Alzforum.
Nonetheless, two significant challenges remain. One persistent concern bruited by many academic researchers is whether BACE inhibitors will prove safe enough for long-term use. Most Phase 1 trials have been brief, meaning that subtle adverse effects might not have had time to develop. A second unknown is whether lowering BACE1 activity will actually slow cognitive decline, especially in people who already have AD. Some researchers suggested these drugs may be more effective as a preventative, and worried that a negative clinical trial in AD could cause companies to give up on the drugs prematurely.
BACE1 Inhibition Effective in AD Patients, Too
Merck previously reported that its BACE inhibitor MK-8931 lowered CSF Aβ by 90 percent or more in healthy volunteers in a short Phase 1 trial (see ARF related news story). In Florence, Forman added data from a seven-day trial of 32 mild to moderate AD patients at three U.S. sites. This is an important step because people with AD, unlike most healthy volunteers, have a head full of amyloid, and that could change their drug response. Participants had a mean age of 73 and Mini-Mental State Examination score of 22, indicating mild AD. They received doses of 12, 40, or 60 mg daily. There were no serious adverse effects, with participants most commonly reporting dizziness, or headache and back pain related to the lumbar punctures, Forman said.
The results were consistent with those seen in healthy volunteers, with CSF Aβ40 and Aβ42 plummeting by up to 84 percent at the doses tested, Forman reported. “That helps us build confidence that we are hitting our target in patients, too,” he told Alzforum. “The major focus of this study was to understand the dose response in patients to help support our dose selection, because we do not have formal Phase 2 data to base dosing on.” Simulations from a disease model Merck built showed that at 12 mg, almost all patients will experience at least a 50 percent reduction in Aβ, and at 40 mg, 75 percent, Forman said.
Merck will take these three doses forward into its Phase 2/3 trial in patients with Alzheimer’s dementia, Forman said. This trial will be the largest yet for a BACE inhibitor, with 200 participants in Phase 2 and 1,800 planned for Phase 3 (see ARF related news story). The company also plans to test its inhibitor in prodromal AD patients, but will wait to see the safety data from the ongoing Phase 2 portion of this current trial, Forman told Alzforum. Other scientists at AD/PD expected that second, prodromal trial to start toward the end of 2013.
The Bright Side: All Signals Go From Early Trials
Eli Lilly’s inhibitor LY2886721 is also currently in a Phase 2 trial of 130 people with AD. In Florence, Patrick May described Phase 1 data he previously presented at the Alzheimer’s Association International Conference 2012 (see ARF related news story). Fourteen days of daily dosing slashed BACE1 activity by 50 to 75 percent, and CSF Aβ42 dropped 72 percent, May said. Robert Lai at Eisai Pharmaceuticals had similar data from a multi-dose Phase 1 study of their inhibitor E2609. As reported at AAIC 2012 (see ARF related news story), a two-week trial lowered CSF Aβ by 50-80 percent.
AstraZeneca did not present trial data in Florence, but Samantha Budd, of that company’s Cambridge, Massachusetts site, told Alzforum that a Phase 1 single-dose study of their inhibitor AZD3293 will wrap up in summer 2013. The company plans to release preclinical data at AAIC 2013 in Boston, Budd said. In Florence, Budd provided a tantalizing tidbit at the satellite imaging pre-conference, reporting that plaques shrank in six-month-old Tg2576 mice treated for one month with another of their BACE inhibitors AZ4217. At this age, the animals are actively depositing amyloid.
“BACE1 [inhibition] has been the Holy Grail since the first description of Aβ release,” May said, expressing a common theme. “Now we have a real shot at testing the amyloid hypothesis with BACE inhibitors.” Forman pointed out that, because this approach targets amyloid production at its point of inception, it should suppress both plaque formation and soluble oligomers. Therefore, the therapy could work regardless of which of these is the toxic entity.
The Dark Side––What Could Go Wrong?
With the new drugs getting into the brain and strongly lowering Aβ, a main concern now for many researchers is whether chronic treatment will lead to side effects. BACE1 cleaves numerous proteins. For many of its substrates, other enzymes can pick up the slack, but for some, BACE1 is the major sheddase, Willem said. One such protein is neuregulin 1, which plays a crucial role in myelination of axons (see ARF related news story; ARF news story; ARF news story). “In animals treated with [Lilly’s BACE inhibitor] LY2811376, we observe robust accumulation of [uncleaved] neuregulin 1,” Willem told Alzforum. However, May said in his talk that Lilly has not seen any reduction of neuregulin cleavage in vivo. Other scientists said that the dose needed to cause neuregulin accumulation exceeds that used in people, but all agreed the issue bears watching.
BACE1 knockout mice are outwardly fine, but upon closer examination show subtle defects in axon guidance (see ARF related news story). They are also more prone to symptoms resembling schizophrenia and to epileptic seizures (see ARF related news story). Additional substrates have been identified in screens by Stefan Lichtenthaler at the German Center for Neurodegenerative Diseases, Munich, and Bart De Strooper at the University of Leuven, Belgium (see ARF related news story). A recent study by De Strooper and researchers in Florida found retinal pathology in one strain of BACE1 knockouts (see Cai et al., 2012), but in Florence, Lichtenthaler reported he could find nothing wrong with the retina in a different strain. One caveat all sources agreed on is that even though the field at large refers to these drugs as “BACE1 inhibitors,” most of the current compounds act about equally potently on BACE2, meaning that BACE2 substrates must also be evaluated.
Researchers said it is difficult to predict how problems with myelination or axon guidance would manifest themselves in a clinical trial. “To be prudent, these companies ought to be doing a whole battery of tests that sample a range of behaviors: cognition, memory, neuropsychology,” suggested Robert Vassar at Northwestern University, Chicago, Illinois. “I would monitor vision and balance as well. Companies should cast a wide net, because we really don’t know what we’re going to see.”
Lichtenthaler agreed that pharmaceutical companies may want to look for a range of neurological and vision deficits. Because BACE1 promotes developmental processes like myelination, and some of these programs can be reactivated for repair after injury or brain trauma, BACE inhibitors might be ill advised for people who have recently fallen or hit their heads, he added. Despite this, Lichtenthaler told Alzforum, “I’m relatively optimistic at this point that the BACE inhibitors will not have too many side effects.” Pharmaceutical inhibitors never completely block protein activity, and would be given late in life. Thus, they should produce milder effects than those seen in knockout mice, which completely lack the protein from birth, he said.
How Much and When?
The question of how much to quell the enzyme looms large. The current compounds achieve up to 90 percent drops in Aβ production, but several academic researchers privately wondered if that might be too much for chronic treatment. “We are far away from fully understanding the impact of a BACE1 block on peripheral and central nervous system functions, but a medical treatment should definitely not result in a complete inhibition of BACE1 activity,” Willem told Alzforum. “Since BACE1 is upregulated twofold in AD, a reduction to its normal levels would be hopefully achievable and probably safe.” Lichtenthaler agreed, “My gut feeling is that if you reach somewhere around 50 percent inhibition, that would already be very good.”
However, Vassar told Alzforum, “Our best guess is that we’re going to have to inhibit at more than 50 percent of BACE, because the heterozygous BACE knockouts still make 90 percent of the normal amount of Aβ. I think we may have to inhibit 70-80 percent of BACE. That still leaves 20-30 percent of BACE around to perform the normal functions. I’m hopeful that we can achieve a therapeutic dose that balances efficacy with safety.”
Last summer’s discovery of a naturally occurring, protective APP mutation has shown that a lifetime dip of about 20 percent in Aβ production significantly protects against AD (see ARF related news story). “That helps us take an educated guess at the key question of how much amyloid reduction you need and how much is safe,” May said. For a treatment started late in life, when people already have plaque in their brains, however, stronger intervention might be needed. Budd suggested, “If we can safely inhibit at higher levels, perhaps that’s good in this late stage of disease.”
Merck’s current Phase 2/3 trial is enrolling people with mild to moderate AD, and they indeed chose doses that inhibit Aβ production by 90 percent. Some researchers worry that this approach could set the drugs up for failure. “I’m skeptical that Merck will see an effect in mild to moderate AD, even at 90 percent inhibition,” Vassar told Alzforum. At that stage of the disease, people already have been losing synapses and neurons for some years. Even if BACE1 inhibition prevents the formation of toxic Aβ oligomers, cognition may not improve, he speculated. Lichtenthaler concurred. “It may be the trial will fail because the patients are not doing much better. But maybe these are really great drugs and it’s simply that they were taken too late. I think [the drugs] would perform much better in prevention trials,” Lichtenthaler said.
Pharma scientists said privately that they suspect Merck knows this and has already decided to keep developing its inhibitor even if patients in the current trial prove too advanced to benefit clinically. Biomarker and safety data to help plan earlier-stage trials, as well as to establish drug effect and safety data across the entire spectrum of this disease, may represent enough gain to run a trial at the stage where, to date, most anti-amyloid drugs have failed (see also ARF Q&A With Merck’s Johan Luthman).
Likewise, Willem believes that a BACE inhibitor alone may do little to lower pre-existing plaque load. The solution may be to use a combination therapy. “After Aβ vaccination in an early stage of the disease, a mild BACE1 block could additionally control Aβ synthesis,” he suggested. Interest in combination therapy is growing in the AD field (see ARF related news story).—Madolyn Bowman Rogers, with reporting by Gabrielle Strobel.
No Available Comments
Read a PDF of the entire series.
Neurofibrillary tangles of tau protein litter Alzheimer’s disease brains, but are they actually bad for neurons? Many researchers now think that soluble, prefibrillar forms of tau are more toxic, and some wonder if tangles could even be neuroprotective. At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March 2013 in Florence, Italy, scientists added evidence for this view by showing that neurons loaded with tangles continue to function in a network circuit. Other speakers described similar data exonerating insoluble tau. The acquittal for tangles does not mean smaller aggregates are off the hook, however, as researchers pointed a finger at tau’s ability to aggregate as its key toxic feature. Others aimed to settle a revived controversy over where tau pathology begins in the brain, and whether some tau deposition is a normal feature of aging.
Insoluble Tau Not the Villain?
Numerous studies have shown that lowering soluble tau can improve memory in animal models even in the presence of neurofibrillary tangles (NFTs) (see, e.g., ARF related news story; Oddo et al., 2006; Berger et al., 2007). Do tangles themselves harm cells? To get at this question, Kishore Kuchibhotla at New York University School of Medicine examined how tangle-bearing neurons function in neural networks of live mice. In Florence, Kuchibhotla presented his recent work, conducted in the laboratories of Brad Hyman and Brian Bacskai at Massachusetts General Hospital, Charlestown, in collaboration with Susanne Wegmann and Tara Spires. He used Tg4510 mice, which express mutant human P301L tau, develop NFTs throughout the cortex, and eventually lose neurons. To visualize neuronal activity, he injected virally encoded Yellow Cameleon, a fluorescent calcium indicator, into layers II/III of visual cortex. He then placed awake animals under a multiphoton microscope and showed them pictures, while observing neuronal responses through a transparent cranial window.
Tangle-bearing neurons in visual cortex maintain their ability to respond to specific line patterns. Shown here are control neurons. Image courtesy of Kichore Kuchibhotla
Kuchibhotla’s preliminary findings suggest that, at a systems level, even neurons jammed with tangles still function. Visual cortical neurons in Tg4510 mice had similar receptive field properties to those in control mice. For example, when the animals looked at striped lines, their neurons fired in response to specific angles, a property called orientation tuning. Kuchibhotla then sacrificed the mice and stained for NFTs to confirm that these functioning neurons contained tangles.
“At a minimum, we can say that tangle-bearing neurons are integrated into the local network,” Kuchibhotla told Alzforum. He is now investigating other neuronal properties, such as the magnitude of the response and how well coordinated neuronal firing is, to see if the cells have more subtle deficits. In future work, Kuchibhotla and colleagues plan to stain for soluble forms of tau in the neurons as well. Then they will turn off tau production in the Tg4510 mice, allowing them to compare cells that contain only tangles, only soluble tau, both, or neither. Ultimately, Kuchibhotla hopes to dissect the contribution of soluble and insoluble forms of tau to cell dysfunction, he told Alzforum.
Catherine Cowan, working with Amritpal Mudher at the University of Southampton, U.K., went a step further in suggesting that insoluble tau could be neuroprotective. In Florence, Cowan described data obtained from a Drosophila tauopathy model (see Cowan et al., 2010). The flies express highly phosphorylated wild-type (0N3R) human tau. They have problems moving, but do not get tangles or neuronal death. This shows that soluble tau alone can cause neuronal dysfunction, Cowan said. When she inhibited kinase GSK-3β in these animals, tau phosphorylation dropped and the flies recovered normal movement. Intriguingly, the treatment also produced insoluble, granular tau aggregates. These electron-dense granules are globular, 20-50 nm in diameter, and contain about 40 units of tau each. Similar granular aggregates have been seen in AD brains at Braak stage I, and are believed to be a precursor to fibrillar deposits, Cowan noted (see Maeda et al., 2006; Maeda et al., 2007). Because the appearance of the aggregates coincides with behavioral improvement, it is possible that these deposits help sequester abnormal, toxic tau and protect cells, Cowan speculated. This implies that treatments that dissolve tau deposits could prove counterproductive.
Soluble Aggregates Could Be Guilty
If not insoluble tau deposits, then what about small soluble aggregates as the toxic forms? Eckhard and Eva-Maria Mandelkow at the German Center for Neurodegenerative Diseases, Bonn, previously developed a strain of mice that expresses a mutant form of tau particularly prone to clump up, and another strain with tau that cannot aggregate. Pro-aggregant mice have learning and memory defects that vanish when tau production is shut off, even though tangles persist. Anti-aggregant mice are fine (see ARF related news story). Would an inhibitor of tau aggregation, such as methylene blue, similarly protect pro-aggregant mice? A derivative of this chemical, dubbed Rember®, is currently in clinical trials for AD and frontotemporal dementia (see ARF related news story; ARF news story). Recent studies have suggested methylene blue works by oxidizing tau, and thus prevents aggregation (see ARF related news story).
In Florence, Eva-Maria Mandelkow reported that 15-month-old pro-aggregant mice that received methylene blue for one to three months showed no cognitive improvement. However, when given the drug from birth, the adult mice performed as well as wild-type animals. The therapy works as a preventative, but not a treatment, Mandelkow concluded. She then looked to see how late in life therapy could be started and still rescue cognition. She found that if she treated pro-aggregant mice with methylene blue for six months, starting at eight months of age and before cognition falters, their memory stayed sharp. Treated animals had fewer prefibrillar tau aggregates and less hyperphosphorylated tau, but more soluble tau. They also had more markers of autophagy, a cellular process that helps dispose of unwanted protein. With this drug, treatment needs to begin before cognitive decline kicks in, she said, adding, “We have to find that point in patients.” Biomarker studies in people who have inherited a familial AD gene show that tau levels rise a few years before clinical symptoms (see ARF related news story).
In contrast to the pro-aggregant protein, excessive amounts of non-aggregating tau seem to have few ill effects. In Florence, Maria Joseph in the Mandelkow's group highlighted the differences in the two mouse strains. Using hippocampal slice cultures from neonatal animals, she saw that slices from pro-aggregant mice had massive pathology and mislocalized tau (see Messing et al., 2013), while those from anti-aggregant mice looked healthy, without hyperphosphorylated tau or microgliosis, and still with all their dendritic spines. Intriguingly, these anti-aggregant slices had an abundance of neurons, more than in wild-type mice. They contained more proliferating cells and more cells that stained with markers of neurogenesis compared to wild-type, suggesting more neurons are being born. Likewise, live anti-aggregant mice sported enlarged hippocampi. Mandelkow told Alzforum she plans to investigate the mechanisms behind this finding.
Where Does Tau Pathology Start?
Current ideas about AD progression propose that amyloid pathology initiates the disease and kicks off tau tangles (see, e.g., ARF Webinar). However, a recent study by Heiko Braak at Goethe University, Frankfurt, Germany, cast doubt on this sequence by reviving an old debate about whether tau pathology comes earlier (see Braak et al., 2011). While classic Braak staging shows AD pathology beginning in the entorhinal cortex, the new work found pre-tangle tau deposits in the brainstem of the majority of people under 30. This has led some researchers to suggest that the disease starts very early in life, with tau pathology in the brainstem. To investigate this issue, neuropathologist Kurt Jellinger at Vienna University School of Medicine, Austria, examined the brains of 239 unselected autopsy cases from ages 55 to 102. Almost half had AD, one-third were healthy control brains, 16 percent had AD with Lewy body pathology, 5 percent had Parkinson’s disease, and 2 percent, dementia with Lewy bodies.
In Florence, Jellinger reported that he did see a subtle tau pathology in subcortical nuclei, such as the locus coeruleus, substantia nigra, and olfactory bulb, in brains at Braak stage zero or I. However, only about half of people at stage I had it. The prevalence gradually rose with increasing Braak stages to reach 100 percent at stage VI. The amount of pathology also correlated with stage, with brains at stage V or VI displaying massive tau tangles in subcortical areas. “This suggests that these regions become increasingly involved during AD progression, rather than representing sites initially affected by AD-associated tau pathology,” Jellinger said (see Attems et al., 2012). This idea is not new. For example, John Morris at Washington University, St. Louis, Missouri, has long held that some modest tau pathology is a normal part of aging (see ARF interview). It is present in almost all older brains regardless of cognitive status. Once amyloid pathology starts, these incipient tangles accelerate and spread, leading to the extensive pathology seen in AD, Morris suggested.
Tackling Tauopathy by Reactivating Plasticity
As a side note, Jellinger’s studies also revealed that neurons surrounded by perineuronal nets containing aggregan seem to be protected against tau pathology. Perineuronal nets consist of negatively charged, sugary proteins called chondroitin sulfate proteoglycans (CSPGs), of which aggrecan is one specific type. The nets form a cage around neurons and help to stabilize synapses and preserve memories. This extracellular matrix develops as critical periods end, preventing further plasticity (for a review, see Wang and Fawcett, 2012; McRae and Porter, 2012). Chewing up the perineuronal net with enzymes has been shown to reactivate plasticity in the adult visual cortex (see Pizzorusso et al., 2002).
Maria Spillantini at the University of Cambridge, U.K., wondered if enhancing plasticity in this way might ameliorate some of the memory defects caused by tauopathies. In Florence, she described work done in collaboration with Michel Goedert, also at Cambridge, and Patrick Aebischer at the École Polytechnique Fédérale de Lausanne, Switzerland. She used a strain of transgenic mice that express P301S mutant human tau and start forming neurofibrillary tangles at five weeks of age, increasing up to four to five months (see Allen et al., 2002). These mice have trouble remembering objects they have seen before, and do worse with age. Spillantini injected chondroitinase ABC, which breaks down the perineuronal net, into six sites in the perirhinal cortex of impaired mice. This brain region is crucial for visual perception and memory. She found that the treatment restored object recognition memory. By contrast, an enzyme that does not digest the CSPGs did nothing for memory. The effect of the treatment was temporary, however. Over the next five weeks the perineuronal net returned and the mice once again became impaired. This serves as a proof of principle that boosting neuronal plasticity might provide a temporary therapy for tauopathy, Spillantini said. Safer methods would have to be found to do it, as this enzymatic approach is not suitable for use in people.––Madolyn Bowman Rogers, with reporting by Gabrielle Strobel.
Read a PDF of the entire series.
Disrupted sleep-wake cycles often accompany neurodegeneration, but scientists are unsure why. When do each of these processes start and how do they influence one another? In the March 11 JAMA Neurology, scientists led by David Holtzman, Washington University School of Medicine, St. Louis, Missouri, report that people’s quality of sleep deteriorates in preclinical Alzheimer’s disease (AD)—when amyloid is depositing but before cognitive symptoms arise. These results suggest the earliest link yet between Aβ pathology and sleep disturbance in humans. Meanwhile, Erik Musiek, a postdoc in Holtzman’s lab, finds that genetically disrupting the circadian clock in mice leads to oxidative stress and damaged neurons. His data point to a relationship between diurnal rhythms and neuronal destruction that is not mediated by Aβ. Musiek presented his results at the 11th International Conference on Alzheimer’s and Parkinson’s Diseases held 6-10 March 2013 in Florence, Italy.
Scientists have long known that people with AD and mild cognitive impairment (see McCurry et al., 1999, and Westerberg et al., 2012) have trouble sleeping. Until now, no one had looked at whether that problems occurs in people with preclinical AD. To find out, Holtzman and colleagues correlated brain amyloid deposition with sleep patterns in cognitively normal people. First author Yo-El Ju and colleagues recruited 142 volunteers with an average age of 65, and observed their sleeping patterns for two weeks. Each participant wore a watch-like device, called an actigraph, to record movement while they slept. The scientists calculated both total sleep time and the percentage of time in bed spent actually asleep, defined as sleep efficiency. Subjects gave a cerebrospinal fluid (CSF) sample within three years of that observation. Based on previous findings (see Fagan et al., 2006, and Fagan et al., 2009) anyone with a value of less than 500 pg/mL CSF Aβ42 was classified as having amyloid deposition in the brain.
Thirty-two people met criteria for likely amyloid deposition. Compared to the rest of the participants, these people slept less efficiently, woke up more often during the night, and trended toward taking more daytime naps. However, total sleep time was similar between both groups. “It suggests that people with amyloid deposition are not getting into deeper stages of sleep as much, and they wake up more frequently,” said Holtzman. More detailed tests will be required to prove that, he added. Impaired sleep efficiency could be an early functional biomarker of disease, since it precedes cognitive decline, wrote the authors.
Which comes first, plaques or sleep trouble? It may be a two-way street, wrote Ju and colleagues. Holtzman’s group previously reported that amyloid deposition precedes sleep-wake problems in APPswe/PS1δE9 mice and that removing brain amyloid restored normal diurnal rhythms (see Roh et al., 2012). Aβ may damage brain regions that control sleep, suggested the authors. On the other hand, the group saw that depriving APP transgenic mice of sleep led to more Aβ in the interstitial fluid while boosting slumber reduced it (see ARF related news story on Kang et al., 2009). Synapses produce more Aβ during wakefulness (see Cirrito et al., 2005) so excessive time awake, resulting in sleep deprivation, could lead to overproduction and a heightened chance of aggregation, the authors reasoned.
Taken together, these findings suggest that sleep disturbances and amyloid may influence each other in a feedback loop, where either can initiate and drive the other. Related factors, such as obstructive sleep apnea and lowered physical activity, likely come into play as well (See figure below).
The interrelationships and positive feedback loops between sleep, β amyloid, AD, and related factors. Figure courtesy of David Holtzman. Copyright (C) 2013 American Medical Association. All rights reserved.
“It confirms a correlation between CSF Aβ levels and some sleep measures,” said Bettina Platt, University of Aberdeen, U.K. However, actigraphy is somewhat inaccurate, she said, arguing that sleep studies require electroencephalography (EEG), which gives more detailed information about stage and depth of sleep. Holtzman agreed and said that he plans to do EEG studies in people with both preclinical dementia and those with mild cognitive impairment. In addition, Platt said “the paper doesn’t offer anything to answer the burning ‘chicken and egg’ question of which [pathology] comes first.” The feedback loop model Holtzman’s group proposed would predict a higher rate of dementia among insomniacs, but no data exists to support that right now, she said. The cause-and-effect relationship is important to understand, because it will imply whether sleep therapy could stave off dementia, whether an erratic slumber pattern marks disease onset and progression, or whether sleep trouble is just another symptom of disease, she said.
While less accurate than EEG, actigraphy did detect a difference between groups and suggests a real relationship between low CSF Aβ42 and sleep efficiency, said Rebecca Spencer, University of Massachusetts, Amherst. This finding supports the idea that sleep fragmentation, more than just total sleep time, is important. Some people may sleep just as much as others but spend more time in light stages, which are less metabolically and cognitively restorative than deep, slow-wave sleep, “That has a lot of cognitive and physical outcomes because people are not getting that restoration,” she said. Spencer is interested in future studies that assess differences in sleep stage in the plaque-depositing population.
The sleep-wake cycle is controlled by the endogenous circadian clock, which regulates many cellular and physiological processes, such as control of free radical damage in peripheral tissues (see Kondratov et al., 2006). Do circadian genes also manage oxidative stress in the brain? To find out, Musiek and colleagues knocked out Bmal1, a master circadian clock gene, to obliterate circadian oscillation in mice. When the mice reached six months of age, the researchers found rampant neuronal oxidative damage, deregulated redox gene expression, astroglial activation, and synaptic damage in their brains. The same thing happened when researchers knocked out other circadian genes.
Interestingly, disordered sleep itself was not the culprit, said Musiek. Conditional knockouts that retained brain Bmal1 expression only in the suprachiasmatic nucleus, the master circadian timekeeper, slept and rose as did normal mice, but had similarly ravaged brain tissue. “This suggested to us that the clock working within neurons and glia is critical, regardless of how the whole animal is functioning,” said Musiek. Perhaps cellular clocks ramp up expression of genes that manage free radicals while the animal is awake, and slow production during sleep, he said. If that clock is disrupted, oxidative stress spirals out of control.
This process could link aging and human neurodegenerative disease, said Musiek. Bmal1 gene expression drops with age and circadian rhythms go out of whack in AD, Parkinson’s and Huntington’s diseases. “If you lose circadian function as you age, you may also lose your defenses against oxidative stress,” he told Alzforum. The researchers will next cross these circadian gene knockouts with APP transgenic mice to see how clock and sleep dysfunction together affect Aβ.—Gwyneth Dickey Zakaib
Read a PDF of the entire series.
Italy’s beautiful city of Florence was the cradle of the Renaissance, a period of rebirth for the arts and commerce that helped Europe shed the comparative crudeness and deprivation of the Middle Ages. It was perhaps inevitable, then, that scientists quipped about Florence as a fitting setting for what they perceive to be a resurgence of the amyloid hypothesis after a beleaguered period of setbacks. On 9 March at the 11th International Conference on Alzheimer’s and Parkinson’s Diseases AD/PD 2013, a plenary session titled “The Amyloid Cascade and Alzheimer’s Disease: Hypothesis or Theory?” filled the venue’s largest auditorium to capacity. Leading scientists recounted why, to their minds, the hypothesis can now be called a theory and is poised on the brink of a more definitive test in the clinic. Some in the audience came away inspired, citing genuine passion on the part of the speakers. Others noted that Lilly’s sponsorship of the symposium—with its slick pocket folder handouts and the pharma company’s oversized logo emblazoned beside the speakers—gave the session an overtly commercial sheen.
In other sessions at AD/PD 2013, Lilly scientists claimed that solanezumab’s small but independently verified Phase 3 benefit for cognition in people with mild AD (see ARF AAN news story) constituted provisional validation of the amyloid hypothesis—a statement that reliably provokes controversy. At the same time, there was a sense at the conference that natural history data, such as those emerging from large groups of people monitored for biomarker change over many years, are swaying the debate in favor of the amyloid hypothesis. The plenary featured a major such paper, published March 7 in Lancet Neurology online from the Australian Imaging Biomarkers and Lifestyle Study of Ageing (AIBL; see Villemagne et al., 2013). AIBL collects clinical, cognitive, neuroimaging, lifestyle, and biomarker data at each visit. “The only valid way of testing the amyloid hypothesis is to understand the natural history of Alzheimer’s disease,” Colin Masters at the University of Melbourne said at the plenary.
In that paper, AIBL researchers describe how they have taken three or more successive measurements, spaced three to five years apart, in 200 AIBL participants across the spectrum of normal cognition to Alzheimer’s dementia. Of the 200 participants analyzed for this paper, 72 had four assessments, and 12 had five assessments covering six years. From those data, the scientists worked out the rate by which amyloid deposition evolves over time and in relation to changes in brain atrophy and cognition. This is the largest longitudinal dataset on how the main manifestations of Alzheimer’s disease change over time before dementia is fully expressed. These data test a proposed staging diagram. AIBL has been running for eight years and is currently entering all 72-month data to refine the rate calculations obtained in the current paper, Masters said.
What did the researchers find? They divided the participants into those who accumulated Aβ over the years and those who do not. Among the "accumulators," cognitively normal people deposit the most amyloid, at a rate of 2.6 percent per year; people with MCI deposit at 2 percent per year; and those with AD at 1 percent per year. The researchers connected the dots by transforming these three- to five-year stretches of true data along the entire spectrum into a cumulative kinetic over time. They found that it takes 12 years on average for a person who is free of amyloid but starting to accumulate it to reach the threshold of positivity in brain amyloid PET, set here as an SUVR of 1.5. Once there, it takes the average person another 19.2 years to have developed Alzheimer’s dementia, defined here as a clinical dementia rating (CDR) of 1.0. “We found the full evolution of this illness takes 30 years,” Masters told the audience.
Importantly, the other markers AIBL tracks—hippocampal and gray matter volume, episodic memory, and other cognitive functions—change much later and faster, within five years prior to this same CDR anchor point.
Sequence of change in the decades-long preclinical development of Alzheimer’s dementia. Image courtesy of Villemagne et al., Lancet Neurology 2013
Since the paper was written, AIBL scientists, including Victor Villemagne, Chris Rowe, and others in Australia, have bulked up the dataset underpinning these timelines. They have added 300 healthy or MCI participants to the imaging cohort in AIBL and another 300 in affiliated studies such as the AIBL-Women's Healthy Aging Study, and the AIBL Veterans Study. They also intend to scan new cohorts for preclinical AD trials to include 300 people. Once more longitudinal scan data are analyzed, “The predictive validity of this curve will become patently obvious,” Masters said.
Other natural history studies are finding much the same thing, though none have as much longitudinal amyloid PET as AIBL. On 5 March 2013, U.S. researchers published similar rates based on two or more serial assessments in 260 participants in the Mayo Study Clinic of Aging (Jack et al., 2013). The studies differ in details, such as how they set their measurement cutoffs, but they converge in their main findings.
Both studies report that for about 15-17 years onward from when a cognitively normal person becomes amyloid positive, the amyloid deposition curve is quite linear, a feature that makes measuring drug effects easier. Also at this AD/PD plenary, John Morris of Washington University, St. Louis, Missouri, pointed to the full decade that passes between when a person deposits significant brain amyloid and when subsequent markers of neurodegeneration and then cognition begin to change. This suggests that in this period of amyloid deposition without serious disruption of synaptic integrity, an anti-amyloid monotherapy may work, but after that, “We may need to use drugs in combination that target more than one mechanism.”
The kinetics of brain Aβ deposition fit a sigmoid curve, with a long, linear stretch in the middle. Image courtesy of Villemagne et al., Lancet Neurology 2013
Both AIBL and the Mayo studies also find that above a certain level of deposition, expressed here as an amyloid PET SUVR of roughly 2.2, the slope levels off and later even becomes biphasic; in other words, the curve dips a bit as dementia advances. In toto, the serial data available to date add up to a sigmoid curve with a long, drawn-out ascent prior to and into the MCI/prodromal stage of Alzheimer’s disease. ADNI finds similar curves, but fewer participants have had serial scans with the same PET tracer to enable calculation of longitudinal rates. ADNI 2 and 3 will generate such data. The first published attempts at quantitatively characterizing longitudinal amyloid deposition, starting from smaller groups and fewer scans, came from John Morris, Mark Mintun, and colleagues at Washington University (Vlassenko et al., 2011; Vlassenko et al., 2012).
Other large natural history initiatives are currently collecting and analyzing serial data on autosomal-dominant forms of Alzheimer’s disease. They have published cross-sectional data of the baseline examination of people at different ages and plotted it to depict extrapolated change over time. These data, too, converge in large part with the true longitudinal data from AIBL, Masters told the audience. In particular, change in Aβ deposition and memory correlate closely between the Dominantly Inherited Alzheimer Network (DIAN) and the AIBL (see also ARF related news story). Curiously, hippocampal atrophy at present looks different, where DIAN appears to find changes earlier than AIBL. “Whether that difference is technical or biological must be worked out,” Masters said.
The data available thus far from DIAN have confirmed that autosomal-dominant and late-onset AD are closely similar in terms of their biomarker and clinical course, Morris said at the plenary. The major phenotypic differences are that motor symptoms, seizures, and amyloid deposition in the basal ganglia occur more often in autosomal-dominant AD than in LOAD (for detailed coverage of DIAN, see ARF related news series). Even as DIAN is continuing its natural history study, with 304 people age 19 and up enrolled and new centers joining, the network has also pushed clinical trials toward an ultimate goal of combination treatments to halt the disease. “DIAN data show that the disease first manifests in overproduction of Aβ. This supports the amyloid hypothesis, but the reason we are doing this is not only to understand the natural history of the disease, but also to test it directly,” Morris said.
To that end, the network created a trials unit (DIAN TU), led by Randall Bateman at WashU. This precompetitive group of 10 pharma companies collaborates on trial planning and nominates their drugs for DIAN trials. Two members, Roche and Lilly, joined a competitive investigation of their unapproved therapies side by side in a single trial, the first such instance in Alzheimer’s disease drug development. Roche and Lilly worked out a contract with WashU in less than a year, agreed on a single protocol for an anti-amyloid preclinical treatment trial, and obtained FDA approval without any protocol changes, as well as IRB approval. “It took less than three months from the announcement of the three selected drugs to our first trial participant signing informed consent on December 31, 2012,” Morris said. That participant subsequently was randomized to receive active drug or placebo on 18 March 2013, officially inaugurating the era of secondary prevention trials in AD, said Morris.
At the AD/PD symposium, Reisa Sperling of Brigham and Women’s Hospital in Boston first noted that many studies around the world are supporting the amyloid hypothesis by suggesting that brain amyloid positivity increases a person’s risk of cognitive decline. AIBL’s March 7 paper demonstrates that by chronicling accelerating decline in people who accumulate brain amyloid but not in those who don’t. The more amyloid a person already has in the brain, the faster this happens. Other studies, such as Sperling’s own Harvard Brain Aging Study, are finding that cognitively normal people who were originally recruited merely to serve as a comparison to the AD groups are turning out to be most interesting. They show amyloid positivity in the same age-dependent fractions that prior pathology studies would have predicted. These amyloid-positive "controls" are now showing subtle cognitive decline in serial assessments. Their age distribution precedes the demographic prevalence of Alzheimer’s dementia by about 17 years. This offers an opportunity for clinical intervention to prevent them from reaching that stage. “We have a 15-year window to act, so I see a glass half full,” Sperling said.
Can therapies prevent red dots from turning into diamonds? AIBL PET data in cognitively normal people show the same age distribution for having amyloid in the brain (red dots) as prior postmortem series (green triangles). This precedes the age distribution for having Alzheimer’s dementia (red diamonds) by about 18 years. Image courtesy of AIBL
But while natural history data—and also genetics (see below)—are increasingly incontrovertible, upcoming clinical trials of the amyloid hypothesis are still groping in the dark on some important questions. That is, in part, because the field remains far from consensus about which toxic species to target, how much amyloid lowering might be right, and when it should be done. Alzforum has closely covered the calls for earlier, preclinical-stage trials prior to extensive neurodegeneration, as well as a budding movement toward combination trials (see ARF A4 news story; see ARF combination trials news series). In Florence, Sperling emphasized that trials need better synaptic markers. For example, in the Harvard Brain Aging Study, functional MRI shows evidence of disconnection or dysfunction of critical networks in amyloid-positive but cognitively normal people; also, their cortex subtly thins out in the precuneus, the posterior cingulate, and other areas that have more pronounced changes later on. These people do worse (Sperling et al., 2013) and decline faster in their thinking than those without amyloid (Lim et al., 2012). For its part, ADNI shows that amyloid-positive cognitive normal people already suffer a slow and subtle cognitive decline (Landau et al., 2012). AIBL shows this, too. “This kind of finding will come to redefine what is normal aging,” Sperling said.
All these studies have large error bars, however. The variation stems in part from cognitive reserve, whereby education and cognitive engagement allow people to withstand the effects of amyloid in their brains for some time. And it stems partly from modifying genes that determine how resilient a person is to Alzheimer’s.
What does genetics say on the topic of the amyloid hypothesis, anyway? Giving a playful nod to Florence, John Hardy from University College London, U.K., borrowed a quip from a book on Leonardo da Vinci: “You can say that genetics has delivered a Dimostrazione for the amyloid hypothesis.” Hardy, whose group discovered the London pathogenic APP mutation and later mutations in tau, α-synuclein, and TREM2, is widely cited for having co-articulated the amyloid hypothesis of amyloid production and downstream tangle pathology (e.g., Hardy and Selkoe, 2002), even though historically, George Glenner first proposed an amyloid-only version of the hypothesis; see Glenner and Wong, 1984. Alas, in subsequent years, Hardy publicly voiced doubt about its explanatory power amid concern that the genetics of late-onset AD failed to adequately support the hypothesis while molecular biology was unable to elucidate the primary function of APP. Those years of uncertainty have now given way to new confidence fueled by technical advances in genetics. In the last few years, GWAS have identified low-risk common variants, exome sequencing has begun turning up high-risk variants, and several large whole-genome sequencing projects are underway.
Importantly, all the genes that GWAS have identified map onto the amyloid hypothesis, said Hardy. They may not affect Aβ levels directly—in fact, in a separate talk at AD/PD 2013, Lawrence Rajendran of the University of Zurich reported that they do not. Yet they fall into the pathways of endosome vesicle recycling, cholesterol metabolism, and innate immunity, all of which intersect with Aβ homeostasis in the complex pathophysiology of AD. For example, the gene TREM2, found last year, reacts to amyloid plaques with enhanced expression, and its protein functions to keep activated microglia in a beneficial phagocytic state, Hardy said. That microglia engulf amyloid has been shown by other groups many years ago, but human genetics validates the relevance of this process in Alzheimer’s disease.
The genetics of Alzheimer’s remains incomplete until new techniques will have accounted for the entire genetic burden of the disease. Even so, the lessons today are that the Mendelian pathogenic Alzheimer’s mutations are all involved in Aβ production, whereas a protective variant cuts the opposite way. GWAS and exome sequencing variants map to defined pathways that are consistent with the amyloid cascade hypothesis. “I feel bullish that we can start to call the hypothesis a theory,” Hardy said.
In discussion with the audience, Sperling pointed out that preventive treatment works in cancer, HIV/AIDS, stroke, osteoporosis, diabetes, and heart disease. She recalled the cholesterol wars, where raging debate about good versus bad cholesterol did not stop the field from running secondary prevention trials in familial hypercholesterolemia (as do DIAN and API), and then thousand-person studies in people thought to be at risk (as does A4). Reduction of cholesterol is now estimated to have reduced cardiac mortality by nearly a third. “For this to work, amyloid does not have to be the cause; it just has to be a critical factor in the cascade,” Sperling said. “We should stop arguing about whether it is amyloid or tau. It is both and it is also other factors that we have not discovered yet. As 10,000 baby boomers are turning 65 every day in the U.S., we need to target everything—amyloid, tau, neuroprotection, metabolism, and the innate immune system.”—Gabrielle Strobel
No Available Comments
Read a PDF of the entire series.
This is Part 1 of a two-part series. See also Part 2.
In a sign of how brain imaging is increasingly infiltrating clinical research on age-related neurodegeneration, the 11th AD/PD International Conference on Alzheimer’s and Parkinson’s Diseases hosted a day-long exchange of new imaging data and discussion of current challenges for the research community in these two disorders. On 5 March 2013, in a renovated space inside the Fortezza da Basso—called “Basilica” for its arched Roman columns, central nave and wings—several hundred researchers met to update each other on the latest in their field. Co-organized by Mike Weiner of the University of California, San Francisco, and Ken Marek, at the Institute for Neurodegenerative Disorders in New Haven, Connecticut, the day consolidated an emerging consensus that in both diseases, outwardly normal people with preclinical imaging evidence of AD or PD face an elevated risk of developing full-fledged disease. This enables researchers to stage people early on. Consequently, a growing number of clinical trials is taking advantage of brain imaging to better define whom to enroll. In contrast, trial designers are struggling with how to use imaging to measure a drug’s effect. More tracers are coming online and are being used on related diseases such as Down’s syndrome. Meanwhile, in the day-to-day work of memory clinics, imaging appears poised—for better or worse, and at considerable cost—to gradually edge out clinical/neuropsychological evaluation in the years to come. “The clinical diagnosis is often wrong. It may become secondary to imaging,” Weiner said. Here are selected highlights.
Chris Rowe of Melbourne University, Australia, shared the latest imaging results from the Australian Imaging, Biomarkers & Lifestyle Study of Ageing. One of the largest longitudinal brain imaging studies around the globe, AIBL engages some 100 scientists and, like ADNI, has brought academia and industry together. Starting in 2006, AIBL researchers ran serial PIB-PET and MRI scans on 288 participants; they have recently increased this number to 750 people.
Prospective clinical studies elsewhere are adding on imaging as well; for example, the Mayo Clinic Study of Aging now runs scans on some 500 of its 2,700 participants. Similar studies include the longstanding Washington University Memory & Aging Project, the Harvard Aging Brain Study, Wisconsin Registry for Alzheimer’s Prevention, Parkinson’s Progression Markers Initiative, as well as De Novo Parkinson, DESCRIPA, and Innovative Medicines Initiative (IMI). They teach researchers how AD and PD develop. For AD, they are testing and refining a popular staging model that is based on hypothetical and cross-sectional data (see ARF related news story).
In Florence, Rowe presented the three- to six-year follow-up from AIBL, the largest batch of longitudinal data to date. He showed how some people accumulate brain amyloid quickly, others slowly, and a third group not at all. The Australian researchers calculated how fast this deposition grows, and how it relates to other markers and symptoms of AD. This work appeared in Lancet Neurology during AD/PD 2013 and was widely discussed at the conference (see ARF companion story). In essence, it takes the average "accumulator" some 12 years to go from a negative scan to the threshold of amyloid positivity, and 19 more years from there to Alzheimer’s dementia. Brain atrophy and memory change much later in the process. When applied to the proposed NIA-AA diagnostic criteria and expressed in their terminology (Sperling et al., 2011; Albert et al., 2011), these data generate a staging diagram with a drawn-out "preclinical" stage of 11-17 years and a five- to eight-year "MCI due to AD" stage. In short, longitudinal AIBL data support new diagnostic criteria that no longer require dementia to make a diagnosis of Alzheimer’s disease, Rowe said.
When fit to new diagnostic criteria, AIBL suggests a preclinical stage of 11-17 years and a mildly symptomatic phase of five to eight years. Image courtesy of AIBL
How can these types of data help doctors formulate a prognosis for individual patients? The field is not there yet, but AIBL is contributing pieces, Rowe said. Following people over time shows that cognitively normal people with brain amyloid decline on repeat testing, whereas amyloid-negative people improve with practice. “Our data support the concept of prodromal or very early AD. People with brain amyloid tend to have poor episodic memory,” said Rowe.
Amyloid positivity is not a simple, binary thing, even though that is largely how such scans may be read in routine clinical practice. In AIBL, the amount of amyloid people had at baseline strongly influenced their likelihood of progressing. Having a lot is more worrisome than having a little. “We think it is important how much amyloid is present, not just whether you are positive or negative on your scan. Being strongly positive is much worse than just being positive,” said Rowe. This finding argues against attaching dire predictions to a barely positive scan. This is one reason why clinical practice guidelines issued this past January argue against offering predictive amyloid scans to cognitively normal people at this time (see ARF related news story).
Because the initial amyloid burden at first scan predicts a person’s progression more strongly than the rate at which it grows, there is no point in serial PET scans for diagnostic purposes. An amyloid scan costs several thousand dollars, and for clinical purposes a second scan generally won’t offer more informative data than the first, Rowe said. Serial MRI scans are a common practice.
Repeat scanning to see how fast people’s amyloid load is growing poorly predicts whether they will be become symptomatic; more can be learned from a single scan measuring exactly how much amyloid they already have. Image courtesy of AIBL
Rowe focused on how amyloid deposition relates to cognitive performance over time. At baseline, differences among the cognitively normal volunteers were difficult to pin down even though the amyloid-positive subgroup did do slightly worse, Rowe said; however, over the next few years, the amyloid-positive people clearly declined. He presented data showing that a cognitively normal people’s odds of meeting diagnostic criteria for MCI due to AD within three years go up 4.8-fold if they have brain amyloid; the odds of progressing from MCI to AD dementia shoot up 14-fold.
Brain amyloid is not harmless. Image courtesy of AIBL
AIBL data are being used to figure out which combination of markers most reliably predicts a person’s likely course over the next few years. Other longitudinal studies are doing this as well. Much of this work currently focuses on people who already are cognitively impaired but still function normally, partly because this MCI stage is where most data exist, and partly because clinical trials are increasingly targeting this population. With regard to who among the MCI cases is on the path to Alzheimer’s dementia, a combination of abnormal markers tends to be more informative than a single marker.
In old story: In Florence, Rowe showed AIBL data suggesting that, for participants who had MCI at baseline, having hippocampal atrophy or carrying ApoE4 ups their odds of dementia within three years by a factor of 4 and 5, respectively. Being impaired on verbal learning and memory boosts that odds ratio to 11, being positive on amyloid PET to 15-fold. The combination of brain amyloid and hippocampal atrophy boosts the odds of developing dementia 44-fold. If a person had brain amyloid and was amnestic, then dementia was virtually guaranteed, with an odds ratio of more than a million, Rowe said.
Other studies are largely convergent. The up to six-year follow-up of the European DESCRIPA collaborative found that its 900 MCI patients seen at 20 clinics across the continent comprised—as did AIBL's—non-progressors and progressors. People who at baseline were abnormal on all three biomarkers measured—in this case amyloid pathology via CSF, brain activity via FDG-PET, and hippocampal shrinkage via MRI—were certain of declining. In essence, they already had AD, just not yet its dementia (partially published Vos et al., 2013; Prestia et al., 2013). ADNI data led to much the same conclusion in a recent analysis of 97 participants. The presence of β amyloid added the single biggest predictive value, but total certainty came from the combination of amyloid, MRI, and an episodic memory deficit, said Murali Doraiswamy of Duke University in Durham, North Carolina (Shaffer et al., 2013).
In Florence, these data sparked discussion about whether clinicians should stop first doing structural MRI scans in people who they suspect have MCI. Should they go straight for an amyloid PET scan? After all, MRI usually requires serial scans to aid prognosis. Some research physicians felt the current practice of MRI first was still appropriate because it can show vascular pathology or other lesions as the reason for the patient’s symptoms. Others suspected that structural MRI for MCI might be on its way out, because many PET scanners in clinical settings can spot tumors and strokes or other lesions, as well.
To Rowe, the convergent progression data mean that MCI is a symptom, not a diagnosis. “But cognitive impairment is what the clinician is faced with, so it is a reasonable group to recruit and follow. With biomarkers, we can pull its heterogeneous causes apart and make real diagnoses,” Rowe said.
Like many groups around the world, AIBL, too, sees a percentage of people with a clinical phenotype of MCI or mild AD whose scan is negative for β amyloid. What do these people have? Their outcomes after three to five years have been dementia with Lewy bodies, the tauopathy supranuclear palsy, frontotemporal dementia, and vascular dementia. Some participants never fit an alternative diagnosis, said Rowe. Some reverted to normal, while others declined until they had to enter a nursing home with an unspecified dementia. Other research groups are finding the same thing, said Doraiswamy, who considers a negative amyloid scan in people with symptoms of MCI particularly useful in the clinic. To parse MCI more completely and predict what will become of the non-AD cases, scientists need tracers for α-synuclein, tau, or the requisite neurotransmitters.
The symposium, as well as the AD/PD conference itself, featured repeated calls for more molecular diagnostics to help clinicians diagnose neurodegenerative diseases more precisely, and to help researchers understand how various pathogenic proteins interact to cause the AD-PD spectrum of illnesses. Postmortem pathology for years has reminded doctors that, at least by the time patients die, many cases of dementia are mixed. Newly issued pathology guidelines have reinforced this message, said Dennis Dickson at the Mayo Clinic in Jacksonville, Florida.
Research on tau tracers is diversifying, with compounds from Japan’s Tohoku University, that country’s Molecular Imaging Center in Chiba, and Siemens Molecular Imaging in early stages of human testing. In addition, Merck is in the middle of a “big effort to screen 3.5 million compounds,” said Richard Hargreaves of that company. On α-synuclein, a Michael J Fox Foundation-funded consortium between researchers at the contract research organization BioFocus near Cambridge, U.K., and U.S. experts including Robert Mach, Bill Klunk, and Chet Mathis has made headway with lead compounds that bind aggregated α-synuclein fibrils; none of them is in human testing yet.
Parkinson’s disease researchers have a molecular diagnostic tool in β-CIT, aka DAT scan. It quantifies the PD-related shrinkage of dopamine transporters in the putamen and striatum areas of the brain. It has been used in trials; however, like amyloid PET tracers, it changes slowly, declining at a rate of about 4 percent per year. “We need imaging markers that show clear change within six months,” said Marek. DAT scans are helping scientists dissect the reasons why people with Parkinson’s get dementia later on (see ARF related news story), and they are helping researchers enrich populations for early-stage trials at the newly redefined premotor and preclinical stages (Stern et al., 2012). Finding such patients is arduous in PD because its incidence is lower than that of AD, said Andrew Siderowf, who recently left the University of Pennsylvania to work for Avid Radiopharmaceuticals, both in Philadelphia.
In Florence, numerous researchers applauded natural history studies such as AIBL. “We should take advantage of the enormous insight we get from longitudinal cohorts, and maintain them as best we can,” said Marek, who leads the PPMI. AIBL is enrolling new people as its advanced patients drop out or die, keeping the study at 1,000 participants; ADNI is currently recruiting for its second phase (see ARF ADNI update story). Industry likes those studies, too, as is evident from their funding of ADNI, AIBL, and PPMI. “We need to follow many subjects examined with biomarkers to autopsy,” said Merck’s Hargreaves.—Gabrielle Strobel.
This is Part 1 of a two-part series. See also Part 2.
No Available Comments
Read a PDF of the entire series.
This is Part 2 of a two-part series. See also Part 1.
At the imaging symposium preceding the 11th AD/PD International Conference on Alzheimer’s and Parkinson’s Diseases, held last month in Florence, Italy, researchers debated how to put insights from longitudinal biomarker studies to use in the next wave of therapeutic trials, which increasingly include costly PET and MR scans.
When drug developers talk about looking for a “sweet spot” in testing their candidate drug, they mean an outcome that changes strongly and in linear fashion so that they can measure what the drug does to it. The natural history studies on AD have reinforced why trialists could sour on the late stages of this disease. As dementia worsens, the rate of amyloid accumulation declines to where it even dips naturally without a drug in the picture. In other words, it becomes biphasic. Biphasic outcomes are notoriously difficult to assess. “You would want to be doing your trials of an amyloid-reducing drug in a cohort where amyloid still accumulates, that is, from an SUVR of 1.5 to 2,” said Chris Rowe of the University of Melbourne, Australia.
Trialists are talking about trying to target drug trials to a "tipping point" in the progression, where people are about to become worse but can still be saved. They also talk about a "critical window," during which anti-amyloid drugs can still affect disease. Most researchers think the 17- to 20-year time span between amyloid positivity and AD represents this window (see Part 1 of this series).
What does this mean in practice? All three secondary prevention trials gearing up in 2013 will use brain imaging. The ADCS’ A4 trial expects to scan some 3,300 people 70 and older for β amyloid and then scan with structural MRI the expected amyloid-positive third among them. To investigate this critical window, the trial will allow—but not require—MRI evidence of neurodegeneration at enrollment and will study post-hoc if the extent of a person’s neurodegeneration affected the outcome, said Reisa Sperling at Brigham and Women’s Hospital, Boston. The A4 trial will stratify people by the extent of their amyloid load, as that appears to influence how quickly they progress (see Part 1) and scan everyone again at 18 and 36 months. The Alzheimer's Prevention Initiative and the DIAN trials of familial AD scan all participants at baseline and 24 months.
This is different from how the bapineuzumab and solanezumab Phase 3 programs used imaging, said Nick Fox of University College London, U.K. For inclusion, both required indirect imaging evidence in the form of an MRI or CT scan consistent with a diagnosis of AD. For outcome, the researchers measured volume changes in various brain areas as secondary endpoints. Both programs differed greatly from the upcoming trials, in that subgroups—not everyone—received outcome MRI scans, and only small subgroups had PET scans. The MRI data have been widely covered for their apparent paradox that the treated group lost slightly more, not less, volume than the placebo group. More analysis since last fall has confirmed that bapineuzumab generated a statistically significant MRI signal, and solanezumab, a trend. The signal consists of a small contraction in the hippocampus, coupled with an expansion of the temporal lobe and of the ventricular space, Fox said. Similar volume changes cropped up with the discontinued active vaccine AN1792 (see ARF related news story).
What does this signal mean? Fox suggested that it might reflect a combination of amyloid removal and fluid shifts that result from changes in associated inflammation. Astrocytes take up a large fraction of gray matter volume, so reduced astrocytosis alone could show up on MRI. This question should be studied, Fox said. Comparing the volumetric response of bapineuzumab with the response to approved antibody treatments in other neuroinflammatory conditions—for example, natalizumab in multiple sclerosis—might be a first step.
Who Is Right—The (Wo)Man or the Scan?
A second pressing research question to come out of the bapineuzumab and solanezumab programs is why a third of the enrolled ApoE4 non-carriers who had amyloid PET scans were negative on them. Call it the October Surprise of Alzheimer’s immunotherapy. This finding last fall startled the trial community. It not only influenced future trials, in which every participant now has to have evidence of brain amyloid by CSF or PET as a condition of entry, but also rippled through the research community with echoes of “we, too.” The same is true in ADNI, AIBL, and local clinics that have begun using florbetapir as a diagnostic aid in uncertain cases.
In fact, at the imaging symposium in Florence, Murali Doraiswamy of Duke University in Durham, North Carolina, said, “We have scanned about 100 patients, and the first word that comes to mind is humbling.” He meant to say that even in cases where expert clinicians felt sure of their early AD diagnosis, the scan came back negative and forced the team to re-evaluate. Thomas Beach of Banner Sun Health Research Institute in Sun City, Arizona, recently published that the clinical diagnoses uploaded to the National Alzheimer’s Coordinating Center database upon autopsy proved to have had a specificity (i.e., had picked the correct disease) of only 70 to as low as 44 percent (Beach et al., 2012). “This means that even leading clinicians are often wrong,” said Doraiswamy.
Philip Scheltens of VU University Medical Center in Amsterdam, the Netherlands, noted that in an ongoing Dutch study of the value of amyloid scans for diagnosis, only 61 percent of the patients who had a clinical diagnosis of AD scanned positive, but so did 80 percent of those with a clinical diagnosis of dementia with Lewy bodies (DLB), and even 28 percent of those the clinician considered to have frontotemporal dementia (FTD). For a fourth of all patients, the diagnosis changed after the amyloid scan.
Scheltens is particularly interested in using amyloid imaging to try to make sense of what ails young patients who present with dementia without a certain cause, i.e., a known mutation. He is conducting the Dutch Flutemetamol in Young Onset Dementia Study. Flutemetamol is G.E.’s 18F amyloid PET tracer; its application for use in clinical practice is pending with the Food and Drug Administration and European Medicines Agency. The study will assess whether an amyloid scan can improve the diagnosis and healthcare management of such cases. The study currently scans four patients per week, and the first images have come back positive thus far, Scheltens said. In the case of a 61-year-old man who complained of progressive memory loss and had hippocampal atrophy, “I myself made an erroneous AD diagnosis,” Scheltens recalled. This man and his similarly affected sister were completely normal on their flutemetamol scan, and later proved to have a tau mutation.
In time, an amyloid scan may change clinical practice by cutting short the remaining assessments. In this study, a third of the clinicians said: “If I have seen the scan, I will not do a neuropsychological evaluation.”
Heading in the same direction, Stephen Salloway of Brown University, Rhode Island, presented data at AD/PD 2013 suggesting that flutemetamol PET scans were more accurate than the clinician had been in a 19-center autopsy study that formed part of G.E.’s marketing application. Among 68 cases of terminally ill older people who agreed to a flutemetamol scan and brain donation, visual up-or-down readings of the scans by five blinded readers matched the postmortem pathology used as the gold standard better than did the clinical diagnosis. In particular, the clinical diagnosis relative to NIA-Reagan Institute pathological criteria came in with a sensitivity of 45 percent and a specificity of 61 percent, whereas flutemetamol clocked a sensitivity of 84 percent and a specificity of 79 percent. “Flutemetamol performed better than the clinical diagnosis,” said Salloway.
Talks like Parkinson’s, walks like Parkinson’s, but isn’t. SWEDDs have something else. Image courtesy of John Seibyl
At least Alzheimer’s researchers are not alone in this problem, said John Seibyl. Their Parkinson’s colleagues, too, see patients in clinical practice and in trials who look like they have PD but scan negative for β-CIT/DAT. This SPECT tracer visualizes loss of dopamine transporters in the basal ganglia and is widely used to support a diagnosis of Parkinson’s. In PD research, these people have a name: "SWEDDs" have Scans Without Evidence of Dopaminergic Deficits (Hall et al., 2010). When researchers dug a little deeper, they found that the scans were right and the clinical diagnosis had been wrong. The SWEDDs turned out to display a "mimic" of parkinsonism that had nothing to do with progressive dopaminergic neurodegeneration. The underlying causes were vascular, a consequence of neuroleptic medication, or a form of tremor. The confusion was big enough for movement disorder specialists in the U.K. to hold a SWEDD meeting in 2010 (see SWEDD newsletter), and PPMI even added a special cohort to compare them side by side with true PD cases and controls (see ARF related news story).
For the completed bapineuzumab and solanezumab trials, the amyloid-negative AD cases remain a riddle. Their investigators are trying to dissect if amyloid-positive and -negative patients responded differently to the therapeutic antibody. Other scientists are wondering if the current PET tracers fail to bind other types of amyloid or if the amyloid-negative people truly did not have AD. For longitudinal studies, Mike Weiner of UCSF suggested that amyloid-negative participants could be offered a DAT scan to see if they have a dopaminergic neurodegeneration masquerading as AD. All clinical cases of DLB have abnormal DAT scans, said Ken Marek at the Institute for Neurodegenerative Disorders in New Haven, Connecticut.
What’s a Trialist to Do?
For ongoing and future clinical trials of anti-amyloid drugs, large numbers of amyloid-negative participants are a huge problem, according to Samantha Budd of AstraZeneca in Cambridge, Massachusetts. “If you want to address amyloid therapeutically, then you want to ensure that amyloid is in the brain,” Budd said. She considers it “certain” that pathophysiology is ongoing years before the current diagnosis, and sees a growing need for imaging diagnostics, given the error rate of clinical diagnoses, especially at the early stages.
Budd spoke from the perspective of a developer of small-molecule drugs who is using imaging to give that drug the best shot in trials. Knowing that enough drug reaches its target in the brain to be able to be effective is critical, but this work sometimes gets short shrift in the race to the clinic. For example, in dissecting the results of the bapineuzumab trials, there is little discussion of the fact that the dose had to be dropped during the trial. “When you drop the dose, you walk away from something that, based on your prior data, you think was necessary,” Budd said, emphasizing how important it is to establish what is the dose and target engagement needed for efficacy.
For this goal, neuroimaging can be helpful. Microdosing—that is, labeling molecules with high-intensity radioactivity—enables scientists to tag a candidate drug, quantify its exposure in the brain, and decide whether to take it forward. If a therapeutic program is able to radiolabel the drug, then non-human primate PET can confirm that the drug gets into the brain sufficiently to support a dose range for human studies. If the program can further generate a PET ligand to the same target, this opens the possibility to look at target occupancy both in non-human primate and in human PET in parallel with the human Phase 1. “Target engagement is fundamental, especially in neuroscience,” Budd said.
In terms of letting only people with brain amyloid into anti-amyloid drug trials, CSF biochemistry and PET are somewhat equivalent, Budd said. Anne Fagan suggested as much in 2006, and subsequent research has confirmed this. As to using biomarkers as outcome measures, none has yet indicated a drug response that heralds a clinical effect; therefore, several candidates need to go into trials at this point. “We need to continue to explore the usefulness of multiple candidate outcome markers,” Budd said.
AstraZeneca is using an amyloid imaging agent it originally developed but in 2011 licensed to Navidea Biopharmaceuticals in Dublin, Ohio, and Andover, Massachusetts. Now called NAV4694, the low white matter binding of this 18F tracer enables it to detect small longitudinal changes and drug effects as sensitively as does PIB (see ARF related news story). Navidea started a multisite, three-year Phase 2b safety and efficacy study in people with MCI this March.
In Florence, Budd said that this tracer could be used to detect a reduction in amyloid as a result of treatment such as with BACE inhibitors (see ARF related news story). Since plaque is an "outcome" of amyloid pathogenesis, a BACE inhibitor should prevent further plaque deposition after some time. This was the case when six-month-old Tg2576 mice, which actively deposit amyloid, were treated with one of AstraZeneca’s BACE inhibitors for one month. “During a consistent reduction of Aβ monomer, we see a reduction in insoluble amyloid,” Budd said. In theory, this is true of solanezumab as well, though the smaller dynamic range and larger white matter binding of the current crop of 18F tracers may render them less able to quantify small changes in amyloid, Budd suggested.
The strategy of testing drugs only in people who have brain amyloid might have one unintended consequence. It may saddle drug developers with having to have a biomarker ready for clinical use as well. “What were those amyloid-negative patients doing in your trials? You need a tool to exclude them. But if you develop a drug that way, then the tool you use to exclude those patients from the trial must be available in clinical practice, too, to not expose those patients to that medicine,” said Cristina Sampaio, a neurologist and regulator who left the European Medicines Agency for CHDI in Princeton, New Jersey, but is still asked to share a regulator’s perspective to the AD/PD field. Some big pharma companies, including Merck, are developing such companion diagnostics alongside their investigational drugs.—Gabrielle Strobel.
This is Part 2 of a two-part series. See also Part 1.
No Available Comments
Read a PDF of the entire series.
8 April 2013. At the 11th AD/PD 2013 Conference held last month in Florence, Italy, the last day unfolded as is often the case at long conferences: Some of the most anticipated talks spilled their new data before sparse audiences. So it was with Bernd Bohrmann’s first public presentation on a preclinical treatment study that combined two active, clinical-stage anti-amyloid drugs. Bohrmann, of F. Hoffmann-La Roche Ltd. in Basel, Switzerland, reported that, when given together, Roche’s Phase 1 BACE inhibitor and its Phase 3 antibody reduced Aβ levels and plaque burden more strongly in a transgenic mouse overexpressing mutant human APP than did either treatment alone. “We can fairly say we saw evidence for additive efficacy,” said Bohrmann.
Researchers from competing companies expressed sentiments that amount to "wish we were doing this." “This is very interesting,” said Dale Schenk, who oversaw development of immunotherapies at Elan and recently founded a new biotech company in South San Francisco called Prothena. “We had an internal discussion about combination therapy,” said Eric Siemers of Eli Lilly and Company in Indianapolis, Indiana, which could conduct similar experiments with its BACE inhibitor and solanezumab or a preclinical anti-plaque antibody published last December (DeMattos et al., 2012). Schenk and Siemers chaired the session. Other scientists privately expressed their interest in this work.
Combination therapy has been a buzzword for some time. Researchers call for it from lecterns and in conversation (see ARF related news story; see ARF news story), while advocacy groups are rallying around it. Regulatory scientists at the U.S. Food and Drug Administration expressly encourage the development of two unapproved, investigational agents (see ARF related news story). Alas, few groups are working on this goal. Among the academics, Philip Wong of Johns Hopkins University has genetically reined in both γ-secretase and BACE activity in mice (Chow et al., 2010). Joanna Jankowsky at Baylor College of Medicine, Houston, Texas, articulated the promise of combination therapy when she throttled Aβ production genetically to hold its levels steady while simultaneously clearing existing plaque with a research vaccine made in Todd Golde’s lab (Wang et al., 2011). Even so, despite widespread acknowledgment that a disease as complex as Alzheimer’s will require multiple hits on goal—either on a single pathway or on several different pathways at once—past talk has been more lip service than action.
In an ARF Q&A, Luca Santarelli of Roche declared an active interest in combination therapy, and in Florence, Bohrmann showed the first data. The BACE inhibitor R7129 reduces both Aβ42 and 40 dose dependently in cell-based assays, CSF, and brain of wild-type and some transgenic mice. The anti-Aβ antibody gantenerumab is more advanced and better known—it binds most strongly to aggregated forms and reduces plaques in people as measured by amyloid imaging (Bohrmann et al., 2012; Ostrowitzki et al., 2011). This antibody is given monthly at doses up to 225 milligrams, and is one of the drugs being tested in the DIAN secondary prevention trial.
Working with Helmut Jacobsen and others at Roche, Bohrmann ran a combination trial of the two medicines in a transgenic mouse by the contract research organization reMYND in Leuven, Belgium. This strain overexpresses the London mutation in human APP. The scientists did not use their in-house mouse models because those express the Swedish APP mutation. That mutation boosts the affinity of the BACE cleavage site on APP, such that the BACE inhibitor was unable to compete well with the mutated APP binding site and the mice responded poorly to the drug. The APP Swedish mutation is extremely rare; most people with Alzheimer’s express normal variants of APP.
In a chronic treatment study, 13.5-month-old—that is, middle-aged mice—with active amyloid deposition received either 30 or 90 mg/kg daily doses of the BACE inhibitor, weekly infusions of 20 mg/kg gantenerumab, or both, for four months. In that time, brain Aβ levels increased sevenfold, Bohrmann told the audience.
BACE inhibition alone reduced Aβ42 and Aβ40 in brain and CSF, and slightly reduced plaque burden. Gantenerumab alone reduced plaque burden and brain Aβ42 but not Aβ40 levels. As expected, the antibody did not change Aβ levels in CSF, as it does not bind strongly to soluble Aβ.
The combination led to much greater Aβ42 reduction and more Aβ40 reduction, Bohrmann said. This effect was driven largely by the inhibitor. The combination also cleared up plaques more strongly; this was driven largely by the antibody. The scientists analyzed total plaque area, as well as change in the number of small, medium-size, and large plaques counted separately, and found that the combination drove amyloid deposition back to below baseline. “No new plaques formed in this mouse model, and small plaques were cleared,” Bohrmann said. This is notable because this mouse model overexpresses the APP London mutation fivefold. “It is a very aggressive amyloidosis model,” Bohrmann said.
Curiously, a residual pool of Aβ40 remained in the treated mice. Scientists debated where that pool is—soluble inside neurons? inside membranes?—and what it means.
Overall, the combination was more effective than either treatment alone. “It makes a lot of sense to go into prodromal trials with a combination like this,” Bohrmann said. “Monotherapy in mild to moderate Alzheimer’s may likely be too little and too late, as was concluded from recent immunotherapy trials by Reisa Sperling.”
For such trials, CSF Aβ can serve as a pharmacodynamic marker for BACE inhibition. The mice responded consistently with advancing amyloidosis, and the researchers saw a CSF signal within two hours of giving the inhibitor. Amyloid PET, even though it may miss some forms of brain aggregated Aβ, still serves as a pharmacodynamic marker of antibody treatment.
Beyond that, however, further mouse studies will tell the scientists little. For example, gantenerumab clears aggregated Aβ via phagocytosis by macrophages/microglia, but this response may be different in humans and mice. Amyloid-induced phagocytosis is more voracious in people than in mice. The current mouse results may be limited in that sense, and human responses should be estimated in an Alzheimer’s disease computational model, Bohrmann said.
Finding the right dosing regimen for human combination trials is the next challenge. For that, too, mice will be of little help. Humans express less APP than these transgenic mice to begin with, and mouse Aβ-degrading enzymes are different. What’s more, rodent metabolism clears drugs faster than in people. For these reasons, scientists generally give mice higher doses. In this study, 30-90 mg/kg was an effective range, whereas in humans, this might end up being the total dose, Bohrmann said. In toto, rodents pharmacokinetics is a poor guide for human trials, and information for dose finding has to come from careful clinical studies, Bohrmann said. Exactly when those can begin depends on Phase 1 results for BACE inhibitor monotherapy.
How about additive versus synergistic? In theory, the hope for combination therapies is for a synergistic effect that neither drug alone can deliver. In practice, however, few combination regimens achieve that, and an additive effect will be good enough for Alzheimer’s, the FDA’s Bob Temple said at a combination trial conference last November (see ARF related news story). “In our mouse study, I would call the result of the combination an additive or enhanced effect. We think that could translate into better efficacy. Whether in humans there is also synergy remains to be seen,” Bohrmann said.
Other combinations, involving anti-inflammatory or anti-tau agents are “open for discussion.” Have the scientists tried an anti-tau antibody together with an anti-amyloid treatment? “Not yet,” Bohrmann said.—Gabrielle Strobel.
No Available Comments
Read a PDF of the entire series.
This is Part 1 of a two-part series on AD/PD 2013 immunotherapy news. See also Part 2.
You could say the concept that endogenous proteins in the body can misfold, aggregate, and spread disease from cell to cell has created infectious enthusiasm for drug development across the field of neurodegeneration and beyond. After all, a pathogenic protein that leaves one cell and passes through intercellular space before slipping into another ought to be a ready target for antibodies to intercept it. If having the “pathogen” spend time outside of neurons does not exactly make it a sitting duck for drugs seeking it out, it does render it more reachable than if it were operating solely from inside neurons. Scientists quickly caught on, and several groups presented new results on this front at the 11th International Conference on Alzheimer’s and Parkinson’s Diseases (AD/PD 2013), held 6-10 March in Florence, Italy.
In a session jointly sponsored by the Swiss biotech company Neurimmune in Schlieren and its larger U.S. partner Biogen Idec in Weston, Massachusetts, researchers showed new preclinical data on antibodies that go after the ALS protein SOD1 and the diabetes protein IAPP (see below). Human antibodies against tau, α-synuclein, and TDP-43 exist but have not been publicly presented yet. In Florence, scientists also presented additional preclinical data on an antibody against the Alzheimer’s protein Aβ and the first clinical results. Taking that story further, scientists from an imaging CRO disclosed first results of how a prodromal AD trial of this antibody is using amyloid PET to test new diagnostic criteria in the imperfect world of multicenter studies (see Part 2 of this series).
Led by Roger Nitsch and Christoph Hock, the Neurimmune scientists have corralled more than 1,000 aged donors, who are either healthy or whose disease is not progressing, into giving blood for analysis of their memory B cells. The idea is that the donors have withstood disease because over their lifetimes, their immune systems have generated and matured effective auto-antibodies against abnormal endogenous proteins. These auto-antibodies may not show up as a straight-up titer in an aged person. Yet they persist in the form of memory B cells and can be amplified by screening these cells against pathogenic protein aggregates in a process the company calls Reverse Translational Medicine. This generated a set of therapeutic human monoclonal antibodies. “We are interested in those that do not bind to physiologic proteins but selectively target the pathologic conformation,” said Nitsch. Jeff Sevigny of Biogen Idec, which licensed several of these antibodies, noted that this makes the antibodies attractive from a safety standpoint. “We think the human immune system is better at engineering antibodies than are people,” said Sevigny.
In Florence, Jan Grimm of Neurimmune added new data to a previously introduced class of antibodies against the enzyme superoxide dismutase 1 (SOD1) as a potential therapy for amyotrophic lateral sclerosis (ALS). While SOD1 mutations account for only a fifth of familial ALS, the protein aggregates in sporadic ALS as well (e.g., Graffmo et al., 2013; Bosco et al., 2010; Forsberg et al., 2010). An ongoing analysis of sporadic ALS tissue samples gathered in Switzerland and Sweden has detected misfolded SOD1 in motor neurons of every one of 20 cases analyzed so far, Grimm said. This was done with Neurimmune’s new SOD1 antibodies. While this is an obvious study to do, it was not possible in the past because antibodies that are highly sensitive for misfolded SOD1 have only recently become available, Grimm said.
This finding makes SOD1 a target for the entire population of some 75,000 patients in Europe, the U.S., and Japan combined, Grimm claimed. It also means that SOD1 remains a drug target, even as newly discovered molecular players have opened up ALS research and researchers are increasingly dividing the disease into different subtypes. “FUS, C9ORF72, and TDP-43 mutation cases have misfolded SOD1, too. It may be a common final pathologic pathway,” Grimm said. SOD1 aggregates can spread from cell to cell (Chia et al., 2010), and adjoining motor neurons degenerate sequentially in ALS (Ravits and Spada, 2009).
In binding studies, one SOD1 antibody specifically targets pathological, misfolded SOD1 with low picomolar affinity but does not bind to misfolded tau or other proteins, Grimm said. The scientists infused 0.1 mg/kg/day of the antibody into the brain in the widely used SOD G93A mouse model starting at 60 days of age, before the mice start having motor symptoms. The chronic treatment almost doubled the number of surviving motor neurons. As the mice got sick, the treated ones retained more weight and a stronger grip, and preserved their gait in a high-speed film assay presented by Tobias Welt at the University of Zurich. The treatment reduced SOD1 aggregate pathology and added 10 days to the mice’s lifespan. This is a significant response in an aggressive model known for its 20-fold SOD1 overexpression. “It is rather difficult to see therapeutic effects in this model,” Grimm said.
The antibody showed a more profound effect in a different model. The G37R line (Wong et al., 1995) overexpresses SOD1 fivefold and develops a slower form of ALS. Weekly injections of 10 mg/kg into the bellies of these mice delayed the onset of symptoms by 49 days, slowed muscle atrophy, and pushed out death by 59 days. “This is the most dramatic effect ever seen in this model,” Grimm told Alzforum, adding that it is also the first reported therapeutic effect with a peripherally delivered antibody in ALS. “These are wonderful data,” said John Trojanowski at the University of Pennsylvania, Philadelphia.
ALS is increasingly understood to also affect cognition. At AD/PD, Fabian Wirth and other researchers in Welt’s laboratory and at Neurimmune reported that they detected SOD1 aggregates in the hippocampus, along with a subtle deficit in object recognition memory—a hippocampal-dependent task. This happened before motor symptoms set in, and antibody treatment reportedly restored this type of memory as well. This experiment was done in the SOD1 G93A model. None of the mouse studies brought up safety concerns, Grimm said.
Therapeutic effects in mice have raised hope before, only to disappoint in subsequent clinical studies (see ARF Webinar; see Shineman et al., 2011). Why would this be different? The new research avoids many of the shortcomings of early preclinical studies that have since been identified, Grimm told Alzforum. It used large groups of up to 24 mice in each experiment, was blinded, and saw the same effect by way of different routes of administration in different mouse strains, whose transgene copy number was confirmed for each study.
Grimm’s antibody is not the only one in play. The Canadian biotech company Amorfix is working with Biogen Idec on antibodies for ALS, and other groups are evaluating antibodies and active vaccines (see ARF related news story).
One Person's Auto-Antibody, Another’s Diabetes Treatment?
From their healthy donors, Neurimmune scientists also derived antibodies against what they consider to be pathologic conformations of fibrillar aggregates of islet amyloid polypeptide (IAPP). This is a peptide hormone implicated in type 2 diabetes. The antibodies do not recognize native IAPP, Nitsch said. Β cells in the pancreas secrete IAPP along with insulin, and IAPP gene expression is co-regulated with expression of insulin in response to glucose. A sugary diet and subsequent insulin resistance hence drive up IAPP production, and when its concentration rises, it forms aggregates that are toxic to pancreatic islet cells. Like Aβ, IAPP is cleaved by neprilysin and IDE.
In a transgenic mouse model that develops IAPP protofibrils and diabetes, 18 weeks of treatment with these antibodies reduced this pancreatic islet amyloidosis. The treatment also improved B cell function. It brought down fasting glucose levels and insulin resistance, and restored the mice’s response to glucose challenge, Nitsch said.—Gabrielle Strobel.
This is Part 1 of a two-part series on AD/PD 2013 immunotherapy news. See also Part 2.
No Available Comments
Read a PDF of the entire series.
This is Part 2 of a two-part series on AD/PD 2013 immunotherapy news. See also Part 1.
At the 11th International Conference on Alzheimer’s and Parkinson’s Diseases, held 6-10 March in Florence, Italy, Thierry Bussiere and Jeff Sevigny of Biogen Idec in Weston, Massachusetts, showed new data on how a therapeutic Aβ antibody their company had licensed from the Swiss biotech Neurimmune was measuring up. The idea is that a fully human antibody derived from old people whose immune systems successfully hold Alzheimer’s disease at bay might be safer than antibodies engineered in the lab (see Part 1 of this series).
Alzforum has previously covered preclinical studies on BIIB037, an IgG1 against a conformational epitope found on β amyloid (see ARF related news story). In essence, BIIB037 binds to parenchymal amyloid and clears parenchymal plaques in transgenic mice without causing microbleeds. In Florence, Bussiere presented new data on a direct comparison of mouse versions of BIIB037 to three other clinical antibodies—bapineuzumab (3D6), solanezumab (m266), and gantenerumab (mGt)—in vitro and in mice.
Both BIIB037 and mGt bound fibrillar amyloid, but the latter bound vascular plaques as well as parenchymal ones, Thierry said. 3D6 bound soluble and fibrillar Aβ equally strongly; among fibrillar deposits, it showed a preference for vascular over parenchymal plaques, while m266 bound only soluble Aβ. Like m266, 3D6 caused a plasma spike due to its binding to soluble Aβ; BIIB037 did not. Nor did BIIB037 cause microhemorrhages in 13-week chronic dosing of 22-month-old mice with doses of up to 70 mg/kg. “We saw a trend for microhemorrhage only at 500 mg/kg—that is a very, very high dose,” Bussiere said. BIB037 starts clearing plaque in mice at 3 mg/kg.
Dosing and safety were the themes of Sevigny’s talk as well. Sevigny presented the first clinical data from an ongoing Phase 1 study comparing six doses of BIIB037 to placebo given as a single intravenous infusion in 56 people with mild to moderate Alzheimer’s. The doses ranged from 0.3 mg/kg to 30 mg/kg. In these studies, it is customary that once the data safety monitoring committee deems a given dose safe, the study proceeds to the next higher dose. Participants came in for assessments at 10 time points up to two years after dosing. To look for amyloid-related imaging abnormalities, aka ARIAs, every participant between baseline and study end underwent four MRI scans, which were read both locally and by a central reader at the imaging CRO Synarc based in Newark, California.
Thus far, 21 study participants had a total of 43 side effects, Sevigny reported. They included headache, diarrhea, and dizziness. Most were mild, some were moderate, none severe, and none became worse with higher doses, Sevigny said. Importantly, no new ARIA-E or ARIA-H came up during the study (some old people and people with AD have ARIA even without drug). Because this antibody seemed safe at the highest dose in this study, the scientists added a test of 60 mg/kg—an improbably high dose that amounts to 4 grams in a 150-pound person. “These are the highest of all therapeutic AD antibodies being tested these days,” Sevigny told Alzforum. The scientists plan to present results of the 60 mg/kg dose later this year.
For comparison, the multiple sclerosis antibody natalizumab (trade name Tysabri®) is generally given as a 300 mg infusion once a month, and the rheumatoid arthritis antibody infliximab (trade name Remicade®) at around 3 to 5 mg/kg.
The pharmacokinetics of BIIB037 was what researchers casually call “well behaved,” meaning it changed in a linear fashion across doses and showed little variability from person to person. Consistent with the preclinical work, the antibody generated no plasma spike, probably because it does not bind soluble Aβ, Sevigny said.
Last summer, Biogen Idec started a second Phase 1 trial to evaluate how people at an earlier stage of disease respond to multiple doses of the antibody. At up to 160 participants at up to 40 locations, this trial is much larger. Besides BIIB037 itself, it also tests whether diagnostic criteria for prodromal Alzheimer’s perform as robustly in the trenches, i.e., in clinics across the country, as they do in the hands of leading clinicians at specialty dementia clinics who articulated them (Dubois et al., 2010; Albert et al., 2011). “We know this diagnosis works in an ideal environment, but a multicenter trial is not an ideal environment,” Sevigny said.
Prospective participants go through a tiered process to gain admission into the trial. People who score between 20 to 30 on the MMSE, between 0.5 and 1 on the Clinical Dementia Rating, and 27 or lower on the Free and Cued Selective Reminding Test (FCSRT) are then referred for an amyloid PET scan with florbetapir and an MRI. The MRI serves to look for vascular lesions and to add anatomic precision to the amyloid scan, but only people whose amyloid PET scan is positive can enter the trial. Trialists across the field are intensely interested in how both the prodromal AD psychometric criteria as well as biomarker enrichment are doing in selecting people who truly are at the earliest symptomatic stage of Alzheimer’s (see ARF AD/PD 2013 story).
Several trials are testing these entry criteria now, and some are beginning to report their initial experience. At the last CTAD conference, researchers talked about a high screen failure rate at sites recruiting for prodromal AD trials (see ARF CTAD story). In Florence, Joyce Suhy of Synarc presented results for BIIB037.
Nineteen imaging centers, using 12 different scanner models from old to state-of-the-art, are participating in the BIIB037 prodromal trial. The sites send the scan data to Synarc. First author Jerome Barakos and colleagues processed them and showed the PET, concomitant MRI, and fused PET/MRI images to the trial’s two central neuroradiologists. These readers were trained on a binary classification method Avid had developed as part of florbetapir’s FDA approval package (see ARF related news story). The scientists also calculated each scan’s SUVR to compare the visual and quantitative methods of interpreting an amyloid scan.
Of the first 80 patients who came to get the scans, 44 had been clinically diagnosed as prodromal AD, with a mean MMSE of 28, and 36 as mild AD, with a mean MMSE of 23. In the visual read, 48 of these 80 people, or 60 percent, were amyloid positive. In the quantitative analysis, 55 were amyloid positive, i.e., concordant with their prior clinical diagnosis.
This means that the visual read had missed seven people who were positive by SUVR, in other words, generated some false negatives. At the same time, however, the comparison of the two methods showed that the visual read produced no false positives, Suhy said. On a technical note, Suhy noted that it’s important to correct the data when a patient moves in the scanner; otherwise, the motion-induced fuzziness in the images can generate a false-positive read. The false negatives were prodromal cases with a mean MMSE of 27 and a composite SUVR between 1.12 and 1.31 (the cutoff for positivity was 1.1). The κ value for how often the two readers agreed was 0.97.
Suhy interpreted the data to mean that enrichment by PET works. “The visual reading scheme allowed accurate detection of all quantitative amyloid-negative patients and their exclusion from this trial,” Suhy said. Around the borderline of amyloid positivity, the visual read missed some people. “We could have had seven more people enter the trial if we had used quantitative analysis, but that would have been at the cost of maybe including a false positive while adding time and process. Avoiding false positives is the main point,” Suhy said. Future multicenter trials, and also clinical practice, will tend to use visual reads because results come back faster and are not dependent on data-processing software.
Of the 32 people who were amyloid negative, a large majority had the prodromal diagnosis. This BIIB037 trial had a higher amyloid-negative rate after the clinical exam than did the bapineuzumab and solanezumab Phase 3 trials. In discussion, scientists said that this is not surprising, because the error rate of clinical diagnosis is generally higher at earlier stages.
Suhy’s talk generated intense discussion about how this enrollment strategy could be improved. Privately, some scientists said that the clinical testing for prodromal AD is far from optimized. In particular, at this point there is considerable uncertainty about how best to set cutoffs for what are, fundamentally, continuous variables.
For his part, Bruno Dubois, at Pitié-Salpêtière Hospital in Paris, suggested that a lower FCSRT cutoff would reduce the number of false negatives. The test’s ceiling is 48, but normative FCSRT data suggest that 27 is the mean for cognitively normal adults. “Maybe half the people in this room, with a little jetlag, would not achieve 27,” Dubois quipped. Dubois uses a cutoff of 17 to make a diagnosis of prodromal AD. “At that point, all our patients are biomarker positive,” Dubois said.
A cutoff of 17 includes only the most impaired end of the MCI spectrum, Dubois acknowledged. Site clinicians said that this number makes recruitment difficult. They spoke about patients whose condition looks like prodromal AD to them but who still score higher than 17 on the FCSRT and therefore never get to have a scan, much less join the trial. On the upside, people who do fall below 17 do not have to be turned away later after having undergone a biomarker test. The basic dilemma is that when the FCSRT cutoff is too high, some people get excluded after an invasive procedure (PET or lumbar puncture); when it is too low, the more mildly impaired people are blocked at the initial clinical step even though they may have a head full of amyloid. Dubois proposed that 20 might be the right compromise.
The BIIB037 prodromal trial cutoff of 27 was set in discussion with Ellen Grober at Albert Einstein College of Medicine in New York. Grober co-developed the FCSRT. The cutoff is higher in part because the U.S. version of the test is different from the French version, for example, using pictures, not words, said Sevigny. By comparison, Roche’s prodromal AD trial of the gantenerumab antibody uses a cutoff of 17. That trial does not permit memantine or cholinesterase inhibitors, whereas the BIIB037 trial does; hence, local factors also influence how a cut point is set.
“The exact clinical criteria for the prodromal trials may have to be tweaked based on first results of ongoing studies. We are all learning,” said Hansruedi Loetscher at Roche.—Gabrielle Strobel.
This is Part 2 of a two-part series on AD/PD 2013 immunotherapy news. See also Part 1.
No Available Comments
Comments
No Available Comments
Make a Comment
To make a comment you must login or register.