Hometown Loyalty: Astrocytes Stay Put During Development, After Injury
Quick Links
Just as people pick up customs and culture from the neighborhoods where they grew up, neurons adopt distinct roles as they develop in certain brain regions. The same appears true for astrocytes, according to a Science paper published online today. Tracking fluorescently labeled astrocytes in transgenic mice, scientists determined these support cells hardly wander from their birthplace, even in response to injury. The findings support the idea that the glial cells develop in local neighborhoods to support particular groups of neurons, and suggest it could be hard to replenish astrocytes damaged by injury or disease. The work was led by co-senior authors David Rowitch and Arturo Alvarez-Buylla at the University of California, San Francisco, and William Richardson at University College London in the U.K.
“This is one of the most important papers ever written on astrocytes,” Ben Barres of Stanford University School of Medicine, Palo Alto, California, wrote in an e-mail to Alzforum. “Its implications are very important, and it shows that we don't begin to understand the roles of astrocytes in normal development and function, and why so much glial heterogeneity is needed.” (See full comment below.) On a similar vein, Barres and colleagues reported in the May 2 Journal of Neuroscience that astrocytes upregulate distinct sets of genes depending on the type of inducing injury (see Zamanian et al., 2012, and associated comments).
Astrocytes support neurons doing all sorts of jobs—some in cognition, others in language or motor control—but it was unclear to what extent the glial cells develop to support these specialized roles. Neuronal diversity arises in part because the brain has hundreds, if not thousands, of small domains from which distinct populations emerge. “We wanted to test whether astrocytes also develop in local neighborhoods, in which case there might be a special relationship between astrocytes and the neurons developing around them,” Rowitch told Alzforum.
First author Hui-Hsin Tsai and colleagues used spatially restricted Cre transgenes to stain specific brain and spinal cord domains of mouse embryos with reporter proteins, then looked in the adult brain for labeled astrocytes emerging from these progenitor domains. For higher resolution, Alvarez-Buylla used Cre-expressing adenoviruses to track smaller populations in the brain. “We found a very precise relationship between where astrocytes end up and where they develop,” Rowitch said. “Their movement is extremely restricted to the place of their birth.” (See image below.) The findings confirm prior work suggesting that astrocytes move in distinct zones during development (Noctor et al., 2004; Schmechel and Rakic, 1979; Voigt, 1989), yet run counter to other reports raising the possibility that the glial cells can travel far and wide (Levison and Goldman, 1993; Windrem et al., 2008).
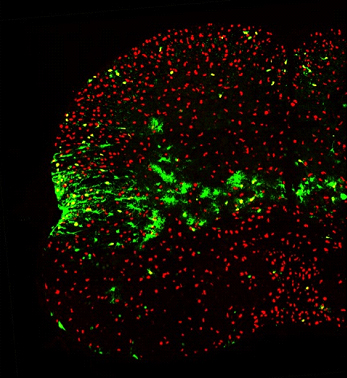
Whereas oligodendrocytes expressing Olig2 (red) spread throughout the spinal cord, fate-mapped astrocytes expressing green fluorescent protein (green) predominantly stay within a restricted area. Image courtesy of William Richardson, University College London
Even if they stay put during development, would astrocytes move to replenish brain areas ravaged by disease or injury? The researchers did two experiments to find out. First, they pierced the spinal cord to create a stab wound, and looked to see if fate-mapped astrocytes came to the rescue from neighboring areas. “They never budged,” Rowitch said. “The only gliotic response was from the local astrocytes.” Second, they depleted astrocytes in specific regions using a suicide gene approach. Again, astrocytes did not migrate in from adjacent domains. Furthermore, the ones left unharmed did not expand to repair and repopulate the area.
“Our data raise concern that the ability for regeneration and repair of astrocyte populations could be extremely limited,” Rowitch said. This may be important for amyotrophic lateral sclerosis (ALS) and other disorders in which astrocytes are damaged. “If we get to the point where we’re transplanting astrocytes in ALS, we might need to transplant the right astrocytes. Our results suggest there may be a specialized astrocyte for motor neurons,” Rowitch said.
Terrence Town of the University of California, Los Angeles, wonders whether astrocytes are similarly restricted in Alzheimer’s disease. “We tend to think of astrocytes being more mobile, surveying the brain milieu and responding to plaques and tangles,” he told Alzforum. “Would AD mice have this same lack of astroglial movement as seen in the acute brain injury models [of the current study]?” asked Town, who called the work "incredibly impressive and thought provoking." Magdalena Götz of Ludwig Maximilians University, Munich, Germany, noted that the ablation experiments “occur very early and may be less relevant to age-related neurodegenerative diseases.” (See full comment below.)
All told, the current work is “a preliminary, fundamental biology study attempting to show how astrocytes may be a very heterogeneous group of cells,” Rowitch said. “This means we should be able to parse them out into different subpopulations during development and during disease conditions to see how some may differ from the resting, supportive population. That’s going to give us new handles on investigating the neuropathology of diseases like AD.”—Esther Landhuis
References
Paper Citations
- Zamanian JL, Xu L, Foo LC, Nouri N, Zhou L, Giffard RG, Barres BA. Genomic analysis of reactive astrogliosis. J Neurosci. 2012 May 2;32(18):6391-410. PubMed.
- Noctor SC, Martínez-Cerdeño V, Ivic L, Kriegstein AR. Cortical neurons arise in symmetric and asymmetric division zones and migrate through specific phases. Nat Neurosci. 2004 Feb;7(2):136-44. PubMed.
- Schmechel DE, Rakic P. A Golgi study of radial glial cells in developing monkey telencephalon: morphogenesis and transformation into astrocytes. Anat Embryol (Berl). 1979 Jun 5;156(2):115-52. PubMed.
- Voigt T. Development of glial cells in the cerebral wall of ferrets: direct tracing of their transformation from radial glia into astrocytes. J Comp Neurol. 1989 Nov 1;289(1):74-88. PubMed.
- Levison SW, Goldman JE. Both oligodendrocytes and astrocytes develop from progenitors in the subventricular zone of postnatal rat forebrain. Neuron. 1993 Feb;10(2):201-12. PubMed.
- Windrem MS, Schanz SJ, Guo M, Tian GF, Washco V, Stanwood N, Rasband M, Roy NS, Nedergaard M, Havton LA, Wang S, Goldman SA. Neonatal chimerization with human glial progenitor cells can both remyelinate and rescue the otherwise lethally hypomyelinated shiverer mouse. Cell Stem Cell. 2008 Jun 5;2(6):553-65. PubMed.
Further Reading
Papers
- Zamanian JL, Xu L, Foo LC, Nouri N, Zhou L, Giffard RG, Barres BA. Genomic analysis of reactive astrogliosis. J Neurosci. 2012 May 2;32(18):6391-410. PubMed.
- Kuchibhotla KV, Lattarulo CR, Hyman BT, Bacskai BJ. Synchronous hyperactivity and intercellular calcium waves in astrocytes in Alzheimer mice. Science. 2009 Feb 27;323(5918):1211-5. PubMed.
Primary Papers
- Tsai HH, Li H, Fuentealba LC, Molofsky AV, Taveira-Marques R, Zhuang H, Tenney A, Murnen AT, Fancy SP, Merkle F, Kessaris N, Alvarez-Buylla A, Richardson WD, Rowitch DH. Regional Astrocyte Allocation Regulates CNS Synaptogenesis and Repair. Science. 2012 Jun 28; PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Ludwig Maximilians University
This is indeed a very interesting paper, as it shows how different astrocytes are compared to oligodendrocytes—the latter migrating far and compensating for any cells that are lost, while the former stay put both during development and in adulthood.
In regard to astrocyte-induced disease, these findings may be relevant, as death of astrocytes in a given domain cannot be compensated for. However, the ablation experiments occur very early and may be less relevant to age-related neurodegenerative diseases.
I think the biggest progress will be to understand the region-specific differences and specialization these cells have, and, hence, understand how they are specialized to support the neurons in their domain.
This is one of the most interesting papers ever written on astrocytes. The implications are very important. Basically, it is showing that each domain of the brain has its own molecularly distinct type of astrocyte, and that these astrocytes respect their own unique boundaries. Most likely this is a very important design plan of the brain. It suggests distinct, domain-specific role(s) for astrocytes. Perhaps they are critical for specificity of axon guidance during development so appropriate neural circuit wiring occurs, as suggested by an earlier paper in Cell by David Anderson a few years ago (see Hochstim et al., 2008). Or perhaps they control domain-specific synapse formation, function, or plasticity. In addition, the paper also shows that killing of astrocytes in one domain results in a substantial decrease in excitatory synapse formation. A role for astrocytes in controlling synapse formation has so far mostly been shown in vitro, so it is very exciting to see evidence that astrocytes also have this role in vivo (see also previous work by Gabriel Corfas on the role of glia in synapse formation in the inner ear: also Rio et al., 2002).
This is a beautiful paper with many important implications, and it shows that we don't begin to understand the roles of astrocytes in normal development and function, and why so much glial heterogeneity is needed. There may be important implications for disease. Perhaps specific types of astrocytes malfunction or degenerate in disease situations. And, since astrocyte gene expression so closely resembles stem cells, perhaps domain-specific types of astrocytes can more easily be induced to become the type of neurons in that domain in order to repair/regenerate lost neuron types.
References:
Hochstim C, Deneen B, Lukaszewicz A, Zhou Q, Anderson DJ. Identification of positionally distinct astrocyte subtypes whose identities are specified by a homeodomain code. Cell. 2008 May 2;133(3):510-22. PubMed.
Rio C, Dikkes P, Liberman MC, Corfas G. Glial fibrillary acidic protein expression and promoter activity in the inner ear of developing and adult mice. J Comp Neurol. 2002 Jan 7;442(2):156-62. PubMed.
Make a Comment
To make a comment you must login or register.