Motor Neuron Vulnerability: The Case of the Missing Chaperone?
Quick Links
What makes motor neurons sensitive to neurodegeneration in amyotrophic lateral sclerosis? Deficiency of a chaperone called macrophage migration inhibitory factor (MIF), suggests a paper in the March 19 Neuron online. MIF prevents mutant versions of the ALS-linked protein superoxide dismutase 1 from misfolding, report senior author Don Cleveland and colleagues at the University of California at San Diego. Adding MIF to cultured neurons prevented mSOD1 toxicity.
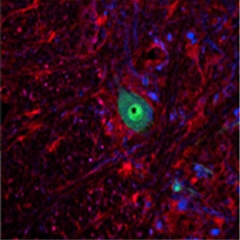
Chaperone AWOL.
MIF (red) occurs in glia, but is conspicuously absent from the motor neurons (green) of a rat spinal cord. [Image courtesy of Neuron, Israelson et al.]
A variety of mutations in SOD1 cause about 20 percent of familial ALS cases. The mutant enzymes fold improperly, and then inappropriately localize to mitochondria and the endoplasmic reticulum (see Jul 2004 news; Jul 2011 news on Vande Velde et al., 2011). Mutant SOD1 also forms aggregates, but it is the mislocalized SOD1, not the aggregates, that instigates disease, explained first author Adrian Israelson, who now works at Ben-Gurion University of the Negev in Beer Sheva, Israel. On mitochondria SOD prevents uptake of ADP, while in the ER it interferes with the degradation of misfolded proteins, inciting a stress response (see Aug 2010 news; Nisitoh et al., 2008).
Since SOD1 is ubiquitously expressed, Israelson wondered why it only mislocalizes to mitochondria and the ER in motor neurons. Those neurons must possess something, or lack something, that creates conditions for mSOD1 to bind these organelles, he reasoned.
To see if the mystery factor was a component of the organelles themselves, or a constituent of the surrounding cytosol, Israelson isolated normal mitochondria from the liver and spinal cord of wild-type rats, and incubated them with cytosol and/or isolated dismutase from mSOD1 rats. Without any cytosol present, the mSOD1 attached to the mitochondria. Liver cytosol inhibited this interaction, but spinal cord cytosol did not. Israelson concluded there was nothing special about the mitochondria, but that the liver cytosol contained a molecule that inhibited mSOD1 binding, and that this molecule was lacking in spinal cord neurons.
To zero in on that unknown actor, Israelson ran the liver cytosol through a size-exclusion column and used mass spectrometry to identify the proteins in the fractions that inhibited mSOD1-mitochondria interactions. This led him to MIF, and he confirmed that purified MIF prevented mSOD1 binding to isolated mitochondria in a dose-dependent manner. He also checked the rat spinal cords for MIF expression, and observed that while it was expressed in glia, it was not present in motor neurons, meaning spinal cords have lower levels of MIF than liver, Cleveland told Alzforum (see image above).
To see if MIF would protect organelles in a whole-cell environment, Israelson co-expressed mSOD1 and MIF in the NSC-34 mouse motor neuron cell line and in human SH-SY5Y neuroblastoma cultures. SOD1 binding to organelles diminished in cultures transfected with MIF.
Because MIF was already known to be a chaperone (Cherepkova et al., 2006), Israelson hypothesized that it might help mutant SOD1 fold. He found it did so in vitro and in the cell cultures. Without MIF, SOD1 accumulated in the neurons and bound an antibody specific for misfolded versions of the dismutase. With added MIF, neurons made similar amounts of SOD1 and almost none was misfolded. Cleveland and colleagues concluded that motor neurons bereft of MIF allow mutant SOD1 to misfold and glom onto mitochondria and the ER.
Next, Israelson used motor neurons derived from the induced pluripotent stem cells of mSOD1 mice to test whether MIF improved cell survival. Normally, these neurons die rapidly, with less than 10 percent surviving five days of culture. When Israelson delivered the MIF gene into the cells via lentivirus, three times as many neurons survived over the same time frame.
Elizabeth Proctor of the Massachusetts Institute of Technology, who was not involved in the study, said she would be curious about MIF’s effects in more complex co-cultures, with glia present. The Cleveland lab had previously discovered that glia expressing mSOD1 attack motor neurons via an unidentified, soluble toxin and that they hasten disease progression, but not onset (see Apr 2007 news; Yamanaka et al., 2008). Israelson and Cleveland plan to test the effects of MIF in co-cultures and in mice.
“This is a remarkable paper, speaking directly to the problem of motor neuron vulnerability,” commented Neil Cashman of the University of British Columbia in Vancouver, Canada, who was not involved in the study (see full comment below). However, absence of MIF may not be a key factor in determining susceptibility, cautioned Piera Pasinelli of Thomas Jefferson University in Philadelphia. The authors have not excluded any other contributors to motor neuron vulnerability, noted Pasinelli, who did not take part in the work. For example, researchers have suggested that the absence of another protein, co-chaperone Sil1, makes the ER of certain motor neurons susceptible to stress (see Jan 2015 news). Israelson also thought it was too soon to estimate the magnitude of MIF’s role in vulnerability.
MIF might have widespread importance, Israelson noted, if scientists are right that wild-type SOD1 misfolds in sporadic forms of ALS (see Oct 2010 news). Pasinelli and Proctor also wondered if MIF might chaperone other proteins that aggregate in the disease, such as TDP-43. “I would like to see that avenue explored,” Proctor said.
In addition to its role as an intracellular chaperone, secreted MIF acts as a cytokine. Israelson hypothesized that extracellular MIF might prevent the prion-like, cell-to-cell spread of defective SOD1 that Cashman and others have proposed (see Dec 2012 conference news). That seems possible, Cashman commented, and would be easy to test.—Amber Dance
References
News Citations
- Motoneuron Mitochondria: Preferred Destination For Mutant SOD1
- ALS: Mutant Enzyme Does Not Hang Around, But Does Find Mitochondria
- New Theory for Some ALS Cases—SOD1 Plugs Cell Power Plants
- Glia—Absolving Neurons of Motor Neuron Disease
- Endoplasmic Reticulum Protein Protects Motor Neurons from ALS
- Research Brief: SOD1 in Sporadic ALS Suggests Common Pathway
- Chicago—ALS Protein SOD1 Painted as Disease Template
Paper Citations
- Vande Velde C, McDonald KK, Boukhedimi Y, McAlonis-Downes M, Lobsiger CS, Bel Hadj S, Zandona A, Julien JP, Shah SB, Cleveland DW. Misfolded SOD1 associated with motor neuron mitochondria alters mitochondrial shape and distribution prior to clinical onset. PLoS One. 2011;6(7):e22031. PubMed.
- Nishitoh H, Kadowaki H, Nagai A, Maruyama T, Yokota T, Fukutomi H, Noguchi T, Matsuzawa A, Takeda K, Ichijo H. ALS-linked mutant SOD1 induces ER stress- and ASK1-dependent motor neuron death by targeting Derlin-1. Genes Dev. 2008 Jun 1;22(11):1451-64. PubMed.
- Cherepkova OA, Lyutova EM, Eronina TB, Gurvits BY. Chaperone-like activity of macrophage migration inhibitory factor. Int J Biochem Cell Biol. 2006 Jan;38(1):43-55. Epub 2005 Jul 26 PubMed.
- Yamanaka K, Chun SJ, Boillee S, Fujimori-Tonou N, Yamashita H, Gutmann DH, Takahashi R, Misawa H, Cleveland DW. Astrocytes as determinants of disease progression in inherited amyotrophic lateral sclerosis. Nat Neurosci. 2008 Mar;11(3):251-3. PubMed.
External Citations
Further Reading
Papers
- Brockington A, Ning K, Heath PR, Wood E, Kirby J, Fusi N, Lawrence N, Wharton SB, Ince PG, Shaw PJ. Unravelling the enigma of selective vulnerability in neurodegeneration: motor neurons resistant to degeneration in ALS show distinct gene expression characteristics and decreased susceptibility to excitotoxicity. Acta Neuropathol. 2013 Jan;125(1):95-109. PubMed.
- Raimondi A, Mangolini A, Rizzardini M, Tartari S, Massari S, Bendotti C, Francolini M, Borgese N, Cantoni L, Pietrini G. Cell culture models to investigate the selective vulnerability of motoneuronal mitochondria to familial ALS-linked G93ASOD1. Eur J Neurosci. 2006 Jul;24(2):387-99. PubMed.
- Kaplan A, Spiller KJ, Towne C, Kanning KC, Choe GT, Geber A, Akay T, Aebischer P, Henderson CE. Neuronal matrix metalloproteinase-9 is a determinant of selective neurodegeneration. Neuron. 2014 Jan 22;81(2):333-48. PubMed.
- Eisen A, Turner MR. Does variation in neurodegenerative disease susceptibility and phenotype reflect cerebral differences at the network level?. Amyotroph Lateral Scler Frontotemporal Degener. 2013 Jul 24; PubMed.
News
- In ALS, “Good” Mitochondrial Protein Turns Partner in Crime
- San Diego: Mutant SOD1 Bumps Mitochondrial Current Up—Or Down?
- No MAM: ALS Protein Breaks Mitochondria-Endoplasmic Reticulum Bond
- Bad Origami: Misfolded SOD1 in Living Color
- Is SOD's Contribution to Lou Gehrig’s Disease Unfolding?
- ALS—Could Stability Rather than Activity of SOD Hold the Key?
Primary Papers
- Israelson A, Ditsworth D, Sun S, Song S, Liang J, Hruska-Plochan M, McAlonis-Downes M, Abu-Hamad S, Zoltsman G, Shani T, Maldonado M, Bui A, Navarro M, Zhou H, Marsala M, Kaspar BK, Da Cruz S, Cleveland DW. Macrophage migration inhibitory factor as a chaperone inhibiting accumulation of misfolded SOD1. Neuron. 2015 Apr 8;86(1):218-32. Epub 2015 Mar 19 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
UBC
This is really a remarkable paper, speaking directly to the problem of motor neuron vulnerability in SOD1 familial ALS and mutant SOD1 mouse models. The authors have systematically sought and identified a chaperone activity in non-neuronal cells that protects from the mitochondrial and ER dysfunction observed with mutant SOD1. That chaperone activity was found to be due to macrophage inhibitory factor. This discovery leads to new and previously unanticipated treatment avenues for SOD1 FALS, and perhaps also sporadic ALS, in which wild-type SOD1 has been observed to be misfolded.…More
View all comments by Neil CashmanUniversite de Montreal, CRCHUM
The study by Israelson et al. identifies a chaperone, MIF, which facilitates SOD1 folding. They report that MIF is of low abundance in motor neurons and might lie at the heart of why SOD1 becomes misfolded and associated with mitochondria. The work is a step forward in our understanding of how mitochondria become coated with and damaged by misfolded SOD1. The findings stimulate many questions. What is the relationship between MIF and the well-characterized copper chaperone of SOD1, CCS? What are the other client proteins of MIF? Is MIF broadly relevant to ALS? Is TDP-43 or FUS protein-folding mediated by MIF, or is it really specific for SOD1? Moreover, what is the broader expression pattern of MIF? There is clearly a deficiency of MIF in motor neurons, but what about other neuronal types? Is MIF relevant to other neurodegenerative diseases where protein misfolding is prevalent?…More
MIF may represent an interesting therapeutic target. The studies presented here are an excellent start toward that goal. It would be ideal to demonstrate that introduction of neuronal MIF is sufficient to rescue disease in the animal model. Given that misfolded SOD1 seems to accumulate more in motor neurons, the lack of MIF may speak directly to disease initiation. However, given that MIF is a cytokine and critical regulator of the innate immune response, I expect that there are challenges ahead.
View all comments by Christine Vande VeldeMake a Comment
To make a comment you must login or register.