Inflammation in Midlife Portends Late-Life Brain Shrinkage
Quick Links
Researchers have long suspected that inflammation fuels neurodegeneration, but evidence in humans has been scarce. A prospective study of more than 1,500 people led by Keenan Walker in Rebecca Gottesman’s group at Johns Hopkins University, Baltimore, adds new evidence in support of this connection. The researchers report in the November 28 Neurology that people with high levels of five inflammation markers in plasma during midlife were likelier to have lost brain volume and memory 24 years later.
- Systemic inflammation in midlife correlates with late-life brain volume loss.
- The occipital cortex and AD-related regions are worst hit.
- Midlife inflammation also predicts memory loss in old age.
“The authors have provided compelling evidence that systemic inflammation not only precedes, but potentially contributes to neurodegeneration in late life,” wrote Angela Jefferson, Vanderbilt University, Nashville, Tennessee, in an email to Alzforum.
Previous studies have uncovered correlations between brain volume and inflammation in older adults (e.g., Jefferson et al., 2007; Schmidt et al., 2016), and between inflammation and cognitive dysfunction, including Alzheimer’s disease (AD) (e.g., Tan et al., 2007; Engelhart et al., 2004). A few have hinted at inflammation driving neurodegeneration, with signs of inflammation surfacing years before mental impairment. Data from a sub-cohort of the Honolulu Heart Program, for example, revealed that men with high levels of C-reactive protein, a nonspecific marker of inflammation, had a threefold higher risk of having dementia 25 years later (Schmidt et al., 2002). Also, a retrospective analysis of data from the U.K. Whitehall II study found that middle-aged people, mostly white men, with high IL-6 blood levels experienced a drop in cognitive function over the next decade (Singh-Manoux et al., 2014; Jul 2014 news).
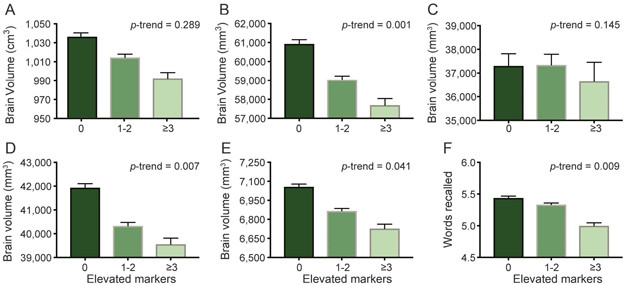
Inflammation and Shrinkage: The number of elevated inflammation markers correlated with smaller volumes of Alzheimer’s-related regions (B), occipital lobe (D), and hippocampus (E). They also correlated with poorer word recall (F). (Changes in total brain (A) and ventricular volumes (C) missed significance in this binned analysis.) [Republished with permission ©2017 American Academy of Neurology.]
But a definitive causal link between midlife inflammation and subsequent neurodegeneration has yet to emerge. To test the strength of this purported link, Walker used the Atherosclerosis Risk in Communities cohort (ARIC Investigators, 1989). ARIC includes people from different communities, with nearly a third being African-American. “It was designed to reflect the general American population,” Walker said. Walker used baseline measurements of systemic inflammation markers in plasma—fibrinogen, albumin, white blood cell count, von Willebrand factor, and Factor VIII—collected from 1,633 participants at a mean age of 53. He then calculated a composite score from these measurements for each person, and assessed its association with brain volume data from MRI scans done 24 years later.
With every increase of one standard deviation in the midlife inflammation score, the combined volumes of Alzheimer’s signature regions—the parahippocampal, entorhinal, inferior parietal lobules, hippocampus, and precuneus areas—shrank by 532 mm3, with the hippocampus alone losing 110 mm3. Participants with three or more elevated markers on average had 5 percent smaller AD signature regions than people with normal inflammatory marker levels (image above). Their occipital lobes and hippocampi were 6 and 5 percent smaller, respectively, than those in people without systemic inflammation. The authors note that the estimated effect of an increase in one standard deviation of the inflammation score on occipital lobe, ventricular, and hippocampal volumes is similar to the effect associated with having one ApoE4 allele.
The authors also searched for links between midlife inflammation and performance in a delayed word-recall test. Asked to remember a list of 10 words, people who had had normal levels of inflammation markers in midlife recalled, on average, 5.5 words. Those with three or more elevated markers recalled, on average, five.
In another recent study, Walker probed the association between midlife levels of the inflammation marker C-reactive protein and white matter microvasculature (Walker et al., 2017). Here, too, midlife inflammation predicted poorer outcomes.
Tom Mosley, University of Mississippi in Jackson, pointed out that midlife blood pressure and midlife diabetes are similarly associated with late-life cognitive impairment (Gottesman et al., 2017; Aug 2017 news). “Taken together, these studies suggest that interventions aimed at preventing late-life brain changes may need to target individuals earlier in life than previously appreciated,” he wrote (full comment below). Mosley’s institution collected data for ARIC, but he was not involved in this study.
How does one stave off inflammation? “Inflammation markers tend to go up with age. They are associated with many diseases, including atherosclerosis, heart disease, obesity, arthritis, and also depression and psychological stress,” said Walker. “All the things the doctor tells you to do to reduce cardiovascular disease can help.”—Marina Chicurel
References
News Citations
- Inflammation in Midlife May Presage Cognitive Decline
- Vascular Problems in 40s, 50s Beget Dementia Down the Road
Paper Citations
- Jefferson AL, Massaro JM, Wolf PA, Seshadri S, Au R, Vasan RS, Larson MG, Meigs JB, Keaney JF, Lipinska I, Kathiresan S, Benjamin EJ, Decarli C. Inflammatory biomarkers are associated with total brain volume: the Framingham Heart Study. Neurology. 2007 Mar 27;68(13):1032-8. PubMed.
- Schmidt MF, Freeman KB, Windham BG, Griswold ME, Kullo IJ, Turner ST, Mosley TH Jr. Associations Between Serum Inflammatory Markers and Hippocampal Volume in a Community Sample. J Am Geriatr Soc. 2016 Sep;64(9):1823-9. Epub 2016 Aug 22 PubMed.
- Tan ZS, Beiser AS, Vasan RS, Roubenoff R, Dinarello CA, Harris TB, Benjamin EJ, Au R, Kiel DP, Wolf PA, Seshadri S. Inflammatory markers and the risk of Alzheimer disease: the Framingham Study. Neurology. 2007 May 29;68(22):1902-8. PubMed.
- Engelhart MJ, Geerlings MI, Meijer J, Kiliaan A, Ruitenberg A, van Swieten JC, Stijnen T, Hofman A, Witteman JC, Breteler MM. Inflammatory proteins in plasma and the risk of dementia: the rotterdam study. Arch Neurol. 2004 May;61(5):668-72. PubMed.
- Schmidt R, Schmidt H, Curb JD, Masaki K, White LR, Launer LJ. Early inflammation and dementia: a 25-year follow-up of the Honolulu-Asia Aging Study. Ann Neurol. 2002 Aug;52(2):168-74. PubMed.
- Singh-Manoux A, Dugravot A, Brunner E, Kumari M, Shipley M, Elbaz A, Kivimaki M. Interleukin-6 and C-reactive protein as predictors of cognitive decline in late midlife. Neurology. 2014 Aug 5;83(6):486-93. Epub 2014 Jul 2 PubMed.
- The Atherosclerosis Risk in Communities (ARIC) Study: design and objectives. The ARIC investigators. Am J Epidemiol. 1989 Apr;129(4):687-702. PubMed.
- Walker KA, Power MC, Hoogeveen RC, Folsom AR, Ballantyne CM, Knopman DS, Windham BG, Selvin E, Jack CR Jr, Gottesman RF. Midlife Systemic Inflammation, Late-Life White Matter Integrity, and Cerebral Small Vessel Disease: The Atherosclerosis Risk in Communities Study. Stroke. 2017 Nov 3; PubMed.
- Gottesman RF, Albert MS, Alonso A, Coker LH, Coresh J, Davis SM, Deal JA, McKhann GM, Mosley TH, Sharrett AR, Schneider AL, Windham BG, Wruck LM, Knopman DS. Associations Between Midlife Vascular Risk Factors and 25-Year Incident Dementia in the Atherosclerosis Risk in Communities (ARIC) Cohort. JAMA Neurol. 2017 Oct 1;74(10):1246-1254. PubMed.
Further Reading
Primary Papers
- Walker KA, Hoogeveen RC, Folsom AR, Ballantyne CM, Knopman DS, Windham BG, Jack CR Jr, Gottesman RF. Midlife systemic inflammatory markers are associated with late-life brain volume: The ARIC study. Neurology. 2017 Nov 28;89(22):2262-2270. Epub 2017 Nov 1 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Mississippi Medical Center
This project identified association between a panel of inflammatory markers, which were assessed in midlife, and regional brain volumes on MRI and memory, both of which were measured 24 years later in life. The findings add another piece of data supporting the negative role that inflammation plays in brain aging and cognitive function.
Given the unique long-term follow-up of the study, it also demonstrates the importance that factors from midlife play in late-life brain changes. Recently, similar results have been reported for midlife blood pressure and midlife diabetes. Taken together, these studies suggest that interventions aimed at preventing late-life brain changes may need to target individuals earlier in life than previously appreciated.
University of Southampton, Faculty of Medicine
University of Southampton
This study measured plasma levels of four acute phase proteins (fibrinogen, albumin, von Willebrand factor and Factor VIII) and white cell count in a mixed-race, middle-age group of 1,633 subjects with later assessment of episodic memory and 3T MRI brain volumetric studies. A composite score of these five inflammatory markers at one time point was shown to be associated with greater ventricular volumes and smaller hippocampi in these subjects 24 years later.
Clearly this study needs to be replicated, but the findings make a lot of sense. Thus, it follows what we know of genetic risk, with the majority of the genetic risk factors in late-onset AD being involved in innate and adaptive immunity (the majority of these risk factors having peripheral as well as central effects). The standard midlife chronic inflammatory markers used in this study are known markers for cardiovascular disease and other inflammatory diseases in later life, which are also known risk factors for AD. So an association makes sense. But is it causal? It would be interesting to assess whether these peripheral inflammatory markers are also detected in the brain, and thus potentially reflecting an impairment or vulnerability of the blood-brain barrier integrity.
The authors mention the relationship between these inflammatory markers and the peripheral cytokines IL6 and TNFα as a possible link with the CNS inflammation. Indeed these links are already in the clinical literature but not mentioned. Thus, Marsland et al.., 2008, have shown that peripheral IL6 levels are associated with smaller hippocampi on imaging in later life. Our own and others’ research shows that systemic inflammation, highlighted by an increase in TNFα, is associated with cognitive decline in AD (Holmes et al., 2009). Blockade of TNFα is associated with reducing cognitive decline in established AD (Butchart et al., 2015) and a reduced risk of developing AD (Chou et al., 2016). These findings and this new study point to causality. Hopefully this study will reawaken interest in peripheral inflammation as a viable treatment target.
References:
Marsland AL, Gianaros PJ, Abramowitch SM, Manuck SB, Hariri AR. Interleukin-6 covaries inversely with hippocampal grey matter volume in middle-aged adults. Biol Psychiatry. 2008 Sep 15;64(6):484-90. PubMed.
Holmes C, Cunningham C, Zotova E, Woolford J, Dean C, Kerr S, Culliford D, Perry VH. Systemic inflammation and disease progression in Alzheimer disease. Neurology. 2009 Sep 8;73(10):768-74. PubMed.
Butchart J, Brook L, Hopkins V, Teeling J, Püntener U, Culliford D, Sharples R, Sharif S, McFarlane B, Raybould R, Thomas R, Passmore P, Perry VH, Holmes C. Etanercept in Alzheimer disease: A randomized, placebo-controlled, double-blind, phase 2 trial. Neurology. 2015 May 26;84(21):2161-8. Epub 2015 May 1 PubMed.
Chou RC, Kane M, Ghimire S, Gautam S, Gui J. Treatment for Rheumatoid Arthritis and Risk of Alzheimer's Disease: A Nested Case-Control Analysis. CNS Drugs. 2016 Nov;30(11):1111-1120. PubMed.
Macquarie University(NSW, Australia), University of Sydney (NSW, Australia), Columbia U (NYC, NY, USA), Pasteur Institute (France), New England Journal of Medicine (NEJM).
This work is a window to expand what we know about the pathophysiology of AD. It is good work and deserves credit. Based on this article and previous findings, I think focus should be directed to:
We now know that factors contributing to blood, bone marrow, and gut cancers are involved in certain neurodegenerative diseases, such as MS and PD. The authors suggested IL-6 involvement in neurodegeneration, as was expected.
There are few studies looking at albumin as a definitive marker for neurodegeneration. It makes sense to consider it as an indirect factor. Looking at Albumin combined with ApoE , LDL, VLDL, cholesterol, and immune factors such histamine would be an interesting path of research.
Enjoyed reading the article and thank you to Alzforum for bringing it to the fore.
Make a Comment
To make a comment you must login or register.