Can a Single Amyloid PET Scan Predict Time to Symptom Onset?
Quick Links
How soon will a person with amyloid plaques in the brain develop the symptoms of late-onset Alzheimer’s disease? In the September 9 Neurology, researchers led by Suzanne Schindler at Washington University in St. Louis debuted a method for answering this question from a single PiB PET scan. How so? Their longitudinal study data showed that once amyloid burden had reached a threshold of 1.2 SUVR, plaques accumulated at the same constant rate in everyone. This allowed the authors to calculate each person’s “amyloid time,” or years since 1.2 SUVR, from one scan. They were then able to predict symptom onset based on a person’s age at the point of crossing that threshold, as older people developed memory problems at a lower amyloid burden than did younger people.
- Above 1.2 SUVR on PiB PET, amyloid accumulates at a constant rate in everyone.
- A single scan can determine a person’s “amyloid time,” or years past threshold.
- Amyloid time can be used to estimate age of symptom onset in LOAD.
Schindler noted that this predictive tool could help researchers select the right patients for clinical trials.
Others were enthusiastic about this work. “This is a great step toward translating the wealth of longitudinal amyloid PET data into a prognostic tool to project possible dementia onset,” Michelle Farrell at Massachusetts General Hospital, Charlestown, wrote to Alzforum. Colin Masters and Victor Fedyashov at the University of Melbourne, Australia, agreed. “The results look very promising,” they wrote, noting the need for replication in larger datasets (full comments below).
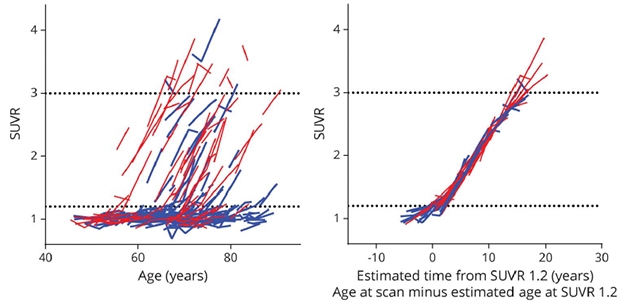
Order From Chaos. Taking each person’s amyloid accumulation trajectory from a spaghetti plot (left) and lining them up using years since a 1.2 SUVR on PiB PET instead of age (right) reveals that plaques grow at roughly the same trajectory in everyone. [Republished with permission, © 2021 American Academy of Neurology.]
Other researchers have reported finding a consistent trajectory of amyloid accumulation in preclinical AD. Sterling Johnson at the University of Wisconsin School of Medicine in Madison correlated amyloid “chronicity,” or accumulation time, with cognitive decline (Oct 2019 news). However, this information had not been translated into a formula for determining age at onset of dementia.
To do this, Schindler used data from 236 people, average age 67, who participated in aging studies at the Knight Alzheimer’s Disease Research Center. All had at least two PiB PET scans, with an average of five years between them, and all but 12 were cognitively healthy at the time of their baseline scan. For amyloid loads below 1.2 or above 3.0 SUVR, the researchers saw little relationship between plaque burden and its rate of accumulation. Between 1.2 and 3.0 SUVR, however, amyloid accumulated at the same steady rate in everyone. There were two phases, with plaques growing rapidly until about 1.8 SUVR, and more slowly thereafter. The SUVR, aka standard uptake value ratio, is a measurement that compares uptake of a radiolabeled tracer in brain regions of interest against that in a reference region not expected to bind the tracer.
Remarkably, when the researchers lined up all the individual trajectories from a spaghetti plot, using time since 1.2 SUVR on the x axis instead of age, the curves became nearly superimposed (see image above). It took 17 years on average to progress from 1.2 to 3.0 SUVR.

Tight Relationship. A formula, based on data from 19 people who progressed from cognitive health to impairment, predicted age at onset within a range of plus or minus three years. [Republished with permission, © 2021 American Academy of Neurology.]
When along this time course do symptoms appear? To try to answer this, the researchers analyzed data from another 180 participants who had at least one PiB PET scan in the 1.2-3.0 SUVR range, and who had undergone annual clinical and cognitive assessments for an average of eight years. The authors found the amount of “amyloid time,” i.e., the time since a person had had an SUVR of 1.2, that elapsed before symptoms showed up varied by age, but not by other factors captured in this study, such as sex, race, education, or even APOE genotype. For example, someone who reached 1.2 SUVR at age 50 would become symptomatic about 20 years later. For a 60-year-old, the average time to onset would be 17 years; for an 80-year-old, 10 years.
“This is consistent with the idea that older subjects may have a greater degree of comorbid pathology, thus a smaller amount of amyloid pathology may be necessary for clinical impairment,” Elizabeth Mormino at Stanford University in Palo Alto, California, wrote to Alzforum (full comment below).
Why did APOE4 have no effect? After all, carriers are known to have a much higher risk of developing AD. Schindler’s data suggest the lipoprotein exerts its effects prior to a person’s reaching 1.2 SUVR. At such low levels of plaque, APOE4 carriers were already accumulating amyloid at a steady rate, while noncarriers were not. The finding jibes with in vitro studies showing that ApoE4 helps seed Aβ, shortening the initial lag phase of aggregation. As a result, APOE4 carriers in these cohorts reached 1.2 SUVR earlier in life, at an average age of 60 years for homozygotes and 64 years for heterozygotes, compared to 69 years for noncarriers.
“A key contribution of [this] paper is the individual-level assessment,” noted Murat Bilgel at the National Institute on Aging in Bethesda, Maryland (full comment below). “Previous studies were able to estimate population-average intervals only.”
Nonetheless, there was some individual variation in the estimated onset age, with a standard deviation of plus or minus three years (see image above). “The influence of genes, environment and age, and the poorly defined concepts of resilience/vulnerability, need to be incorporated,” Masters and Fedyashov wrote.
In future work, Schindler is applying the same methodology to develop predictive formulas for other biomarkers. She noted the method works for the more commonly used tracer florbetapir as well, although the data are not as clean, perhaps because the tracer is less sensitive than PiB. Schindler also has preliminary evidence that the approach can estimate onset age from fluid biomarkers such as cerebrospinal fluid Aβ42/Aβ40.—Madolyn Bowman Rogers
References
News Citations
Further Reading
News
Primary Papers
- Schindler SE, Li Y, Buckles VD, Gordon BA, Benzinger TL, Wang G, Coble D, Klunk WE, Fagan AM, Holtzman DM, Bateman RJ, Morris JC, Xiong C. Predicting Symptom Onset in Sporadic Alzheimer Disease With Amyloid PET. Neurology. 2021 Nov 2;97(18):e1823-e1834. Epub 2021 Sep 9 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Massachusetts General Hospital
This is a great step toward translating the wealth of longitudinal amyloid PET data collected for research into a prognostic tool to project possible dementia onset in currently healthy older adults.
It provides a solid base framework to build upon, as there are many factors (cognitive/neural reserve, cardiovascular health, comorbidities, etc.) that might impact how rapidly an individual may progress from amyloid positivity to clinical symptoms. There is a great deal of potential here, and I look forward to seeing the authors expand on this framework.
I would love to see this data transformed to the Centiloid scale for better generalizability across tracers and scanners. It would also be very exciting in the future to see how well these “amyloid time” PET-based frameworks might correspond with more accessible and affordable plasma biomarkers.…More
University of Melbourne
University of Melbourne
This paper is a most welcome contribution to the critically important goal of developing prognostic algorithms for predicting time of onset of symptoms of AD. The authors use a reasonable number (n = 236) of subjects from the longitudinal cohort of the Knight ADRC who had been followed for an average of five years, and had had more than one Aβ PiB-PET scan. Onset of symptoms was defined by CDR of 0.5 or greater.
The authors draw on a mechanistic model of Aβ aggregation in which a lag-phase is followed by a growth phase driven by nucleation events. Superficially, this is very attractive. Alas, we now know that nucleation is a complex affair, with primary nucleation leading to a pathway of Aβ fibril elongation and secondary nucleation with a pathway toward Aβ oligomerization (Aβo). The former may result in extracellular Aβ plaques and the latter may be the cause of synaptic damage, and tau aggregation and phosphorylation. If the PiB-PET signal reflects mainly primary nucleation events leading to Aβ plaques, it could mean that valuable information is missing if the secondary nucleation events leading to toxic Aβo are not being captured. As we move forward with biofluid (CSF and plasma) analyses, changes which occur before or independent of the PET signal may prove extremely important in deriving the prognostic algorithm.…More
Be that as it may, the authors define a “tipping point,” analogous to the end of the lag phase of primary nucleation, based on their assessment of the first discernible change in the PiB-PET signal [SUVR 1.2, 7 Centiloids (CL)]. Given that the current lower threshold for clinical use is somewhere between 15-20 CL, this tipping point is very low, but still is entirely plausible if it can be confirmed in further studies. The upper-level threshold (SUVR 3.0, 88 CL) is also low, given that 100 CL is by definition the mean full-blown AD dementia level. Nevertheless, the 7-88 CL range is a useful start for beginning the statistical analysis of time of onset.
Methodologically, the paper constructs a disease-progression model based solely on Aβ PET information, called "amyloid time." The idea has intuitive appeal—indeed, it represents how long it has been since one crossed a particular threshold, while taking into account the fact that the rate of accumulation is not constant across Aβ levels.
All studies utilizing similar approaches follow a two-step procedure. First, individual slopes are estimated based on longitudinal data (here per-subject linear regression, but mixed models have been suggested) and are assumed to correspond to the estimated burden at midpoint of follow-up. Secondly, a population-level relationship between current levels and rates of change is examined. Despite being standard, these two steps hold somewhat contradictory assumptions, the former postulating linearity (and thus constant rate of change) at the patient level, the latter studying nonlinear association that emerges once the data have been aggregated.
Perhaps, going forward, and making use of larger longitudinal studies, a better approach would be to explicitly assume that the dynamics of Aβ accumulation are governed by an ordinary differential equation—or, more likely, a mixture thereof corresponding to whether one is a "progressor" or not, and incorporating effects of ApoE4 coming from some parametric family, and subsequently perturbed by measurement noise. The resulting model can then be fitted using probabilistic programming frameworks, e.g., Stan, Turing.jl etc.
As far as predicting age of onset is concerned, the results look very promising. However, to be fully convincing, external validation is required, as the authors themselves acknowledge. The outcome of testing this model in a separate dataset of PiB-based Aβ measurements would be extremely important. A study of robustness with respect to various modelling choices, e.g., bins, knot positions, etc., could also be beneficial.
As a minor point, we note that the estimate of "age at SUVR 1.2" may potentially use information unavailable at the onset of symptoms (if, say, more PET scans were done post-onset) due to the averaging procedure employed, only including observations with SUVR>1.2 that occur prior to onset would alleviate this issue. Showing distribution of "age at SUVR 1.2" estimates per individual coming from different time points is of independent interest and would help gauge the model’s consistency at the personalized level.
Finally, the influence of genes, environment, and age, and the poorly defined concepts of resilience/vulnerability, need to be incorporated. ApoE haplotypes and current GWAS hits are primarily “age at onset” genes, of which ApoE4 is clearly the major co-dominant risk factor. But the genes that determine “rates of progression” are still poorly defined. We and others have confirmed the polymorphic BDNF gene plays a significant role in rates of progression from preclinical to prodromal stages. This type of genetic influence also needs to be incorporated in any prognostic algorithm. Environmental factors, particularly manifest through cerebrovascular co-morbidities, together with other age-related neurodegenerative conditions (such as TDP43–related FTD), also need to be factored in.
Our AD field has indeed reached a “tipping point” in which new improved technologies of neuroimaging, biofluid analyses, genetics, and the cognitive neurosciences will all soon deliver useful prognostic algorithms.
Stanford University School of Medicine
This work adds to a growing literature examining the amount of time an individual has been amyloid+, referred herein as Amyloid Time and referred to as Amyloid Chronicity by Koscik et al., 2020.
I love this approach. It is intuitive and provides an important dimension to explore regarding amyloid burden in our studies. In particular, can a duration measure provide additional information above and beyond simply characterizing individuals as A-/A+? Further, are there factors that modify the impact of amyloid duration?
The work by Schindler suggests that the impact of duration is modified by chronological age, such that individuals with a younger onset of amyloid positivity can bear that burden for longer than their older counterparts. This is consistent with the idea that older subjects may have a greater degree of comorbid pathology, thus a smaller amount of amyloid pathology may be necessary for clinical impairment. However, it is potentially at odds with early onset AD, which tends to have a faster progression. The predictive value of amyloid burden as a function of age will require larger datasets, and potentially progression outcomes that are not as focused on typical presentations of AD (for instance, the initial deficits in younger cases may be non-amnestic).…More
Somewhat related to the comment above regarding early age of onset of amyloid+, I find the distribution of age of onset in Figure 2D among the APOE noncarriers very exciting. This plot highlights that a decent chunk of noncarriers became amyloid+ between age 50-55. This group in particular could be extremely insightful to discover novel drivers of amyloid deposition.
References:
Koscik RL, Betthauser TJ, Jonaitis EM, Allison SL, Clark LR, Hermann BP, Cody KA, Engle JW, Barnhart TE, Stone CK, Chin NA, Carlsson CM, Asthana S, Christian BT, Johnson SC. Amyloid duration is associated with preclinical cognitive decline and tau PET. Alzheimers Dement (Amst). 2020;12(1):e12007. Epub 2020 Feb 13 PubMed.
National Institute on Aging
A key contribution of this paper to the literature is the individual-level assessment of the interval between the onset of elevated amyloid levels and the onset of clinical symptoms of sporadic Alzheimer’s disease. Previous studies that addressed this question relied mainly on datasets where the age at onset of elevated amyloid and the age at onset of symptoms were not both known (or inferable) for each individual. Because of this, previous studies were able to estimate population-average intervals only.
This paper confirms the estimated intervals from these previous studies, and expands upon them by demonstrating that the interval between the onset of elevated amyloid levels and onset of clinical symptoms varies substantially across individuals (as illustrated by the left-most red dot in each row in Fig 3A). As shown by Schindler and colleagues, a factor that explains a substantial proportion of this variability is the age at onset of elevated amyloid levels. Compared to individuals who developed symptoms earlier in life, individuals who developed symptoms later also had developed amyloid later (Fig 3C and 3D). Interestingly, however, those with a later onset of elevated amyloid had a shorter time between amyloid onset and symptom onset compared to those who developed elevated amyloid earlier in life. …More
It is important to remember that not every individual who has elevated amyloid levels develops cognitive impairment or dementia. This makes it difficult to obtain unbiased estimates of the interval between onset of elevated amyloid levels and onset of symptoms. The reported interval estimates in this paper are for only the individuals who did go on to develop impairment. Because of this, the findings of this paper may not be generalizable to every individual with elevated levels of amyloid. Further analyses are necessary to provide insight into who among the amyloid accumulators might develop future cognitive impairment.
Nevertheless, the strong correlation observed between the age at elevated amyloid onset and age at symptom onset suggests that the former might be a promising outcome measure in clinical trials targeting the preclinical stage of Alzheimer’s disease.
Lund University
In a beautifully outlined paper, the authors conclude that at around 1.2 SUVR PiB PET, a fairly steady rate of amyloid accumulation kicks in. After that, it takes about 17 years to reach 3.0 SUVR. This steady accumulation rate toward reaching a specific amount of amyloid burden gives an interesting insight into the pathophysiology of AD.
So does the finding that a predictable rate (correlation between SUVR and accumulation rate) exists in APOE4 carries, but not in noncarriers, below 1.2 SUVR.
The identified cutoff at 1.2 SUVR for PiB PET could be very useful in several settings, ranging from primary prevention trials to studies on preclinical AD, if replicated (preferably using transformed cutoffs for other amyloid PET tracers). The paper is slightly technical at times, but for readers looking for raw data I recommend Fig. 2E, which captures the essence without modeling.…More
Regarding the part about estimating the age of symptom onset, I am a bit hesitant about the generalizability when considering the following: 1) The diagnostic assessment, where clinicians were blinded to both amyloid PET and the assessment at other visits, must have been a challenging task and naturally contains diagnostic uncertainty. 2) These models were mostly derived from small subsamples. 3) Age at symptom onset partly depends on one’s level of cognitive reserve. The present population was much more highly educated (mean years of education of around 15-16) than the average elderly population. 4) They tended also to be somewhat younger than the typical preclinical/early symptomatic AD population (mean age 66 years) and a typical older population often has more co-pathologies that could affect the estimated time to symptom onset.
The approach and effort are nonetheless extremely important, since these kinds of predictions of symptom onset may be very valuable when deciding whom to treat with anti-amyloid drugs in the future (i.e., preferable those with preclinical AD expected to develop symptoms within the next couple of years and not the 45-year-old example case in the paper with an amyloid PET of 1.2 SUVR who was predicted to develop symptoms in 21 years).
Mayo Clinic
There has been a lot of interest in using amyloid age as the time scale. Terry Therneau in our group recently published his algorithm based on the same general idea, see, for example, Fig. 2 in Therneau et al., 2021.
How to line different people up on a common x axis was identified as a problem for AD disease modeling a long time ago. We discuss this in Jack et al., 2010. Schindler et al. have done a nice job with their approach, and their results are believable.
References:
Therneau TM, Knopman DS, Lowe VJ, Botha H, Graff-Radford J, Jones DT, Vemuri P, Mielke MM, Schwarz CG, Senjem ML, Gunter JL, Petersen RC, Jack CR Jr. Relationships between β-amyloid and tau in an elderly population: An accelerated failure time model. Neuroimage. 2021 Nov 15;242:118440. Epub 2021 Jul 29 PubMed. …More
Jack CR, Knopman DS, Jagust WJ, Shaw LM, Aisen PS, Weiner MW, Petersen RC, Trojanowski JQ. Hypothetical model of dynamic biomarkers of the Alzheimer's pathological cascade. Lancet Neurol. 2010 Jan;9(1):119-28. PubMed.
University of Wisconsin-Madison
This study is an important addition to our field for all the reasons others have commented on so far. I want to draw attention to the “order from chaos” plot in the Alzforum article, which is from Figure 2 in the paper. Everyone who is amyloid-positive is accruing signal at roughly the same rate (within measurement error). The heterogeneity is largely in the time domain (age at which the trajectory crossed the x-axis and became positive).
This is consistent with group observations from Villemagne et al., 2013, and several others, including recent work from Jagust and Landau, 2021, that amyloid accumulation PET signal is predictable.
From our work in Koscik et al., 2020, on the Wisconsin Registry for Alzheimer’s Prevention, and the updates we showed at AAIC 2021, and now this important paper from Schindler et al., we can be reasonably confident not only that it's linear, but also that the rate of change is largely the same for all the A+ participants in the plot. Bilgel et al., 2016, were the first to show some of these ideas at the individual level in the BLSA cohort. As was demonstrated nicely by Schindler et al., this is going to enable individual-level estimates of age of onset from a single PET scan! This should greatly inform our understanding of the biomarker cascade and may be informative clinically.…More
I also want to point out an important implication of these results. The rate of amyloid change in A+ subjects looked to be about the same for ApoE4+ and ApoE4-. Similarly, we showed at AAIC 2021 that the rate of change in A+ subjects looked the same for men and women, and for people who got on the amyloid time escalator at younger or older ages. We can reasonably hypothesize, based on the curves we are seeing, that since rate of change is impervious to these potent non-modifiable risk factors, it is likely also impervious to modifiable risk factors. This is now testable.
Also, as we begin to more broadly apply this class of methods and extract the temporal information from amyloid PET in cohort studies, we can begin to actually quantify the many factors that might influence cognitive decline, anchored to person-level amyloid onset age. This new capability will greatly help our field understand vulnerability and resilience to cognitive decline and conversion to clinical AD stages, anchored to amyloid time.
References:
Bilgel M, An Y, Zhou Y, Wong DF, Prince JL, Ferrucci L, Resnick SM. Individual estimates of age at detectable amyloid onset for risk factor assessment. Alzheimers Dement. 2016 Apr;12(4):373-9. Epub 2015 Nov 14 PubMed.
Jagust WJ, Landau SM, Alzheimer's Disease Neuroimaging Initiative. Temporal Dynamics of β-Amyloid Accumulation in Aging and Alzheimer Disease. Neurology. 2021 Mar 2;96(9):e1347-e1357. Epub 2021 Jan 6 PubMed.
Koscik RL, Betthauser TJ, Jonaitis EM, Allison SL, Clark LR, Hermann BP, Cody KA, Engle JW, Barnhart TE, Stone CK, Chin NA, Carlsson CM, Asthana S, Christian BT, Johnson SC. Amyloid duration is associated with preclinical cognitive decline and tau PET. Alzheimers Dement (Amst). 2020;12(1):e12007. Epub 2020 Feb 13 PubMed.
Villemagne VL, Burnham S, Bourgeat P, Brown B, Ellis KA, Salvado O, Szoeke C, Macaulay SL, Martins R, Maruff P, Ames D, Rowe CC, Masters CL, Australian Imaging Biomarkers and Lifestyle (AIBL) Research Group. Amyloid β deposition, neurodegeneration, and cognitive decline in sporadic Alzheimer's disease: a prospective cohort study. Lancet Neurol. 2013 Apr;12(4):357-67. Epub 2013 Mar 8 PubMed.
Make a Comment
To make a comment you must login or register.